By
Charlotte Simmler, PhD; Shao-Nong Chen, PhD; Jeff Anderson, MS; David C. Lankin,
PhD; Rasika Phansalkar; Elizabeth Krause, PharmD; Birgit Dietz, PhD; Judy L.
Bolton, PhD; Dejan Nikolic, PhD; Richard B. van Breemen, PhD; and Guido F.
Pauli, PhD
UIC/NIH Center for Botanical Dietary Supplements Research
College of Pharmacy, University of
Illinois at Chicago
Chicago, Illinois
What are Botanicals? Raw materials, ingredients, and
products derived from plants are commonly referred to as herbs or botanicals in both the biomedical
literature and the natural products health industry. This overarching term
includes the breadth of crude herbs, plant parts, and the ingredients made from
them, and also covers finished products such as botanical dietary supplements. Botanical dietary supplements are intended
to supplement the human diet and are composed primarily of powdered plant
parts, their extracts, or other preparations derived from crude herbal material;
some formulations include other ingredients such as vitamins, minerals, and
amino acids. Botanical dietary supplements are highly complex mixtures
reflecting the diverse chemical constituents that comprise the source plant’s
raw material. Botanical analysis is an intricate analytical challenge requiring
specialized skills and instrumentation that is different from those required
for quality control of chemically simpler pharmaceuticals, or for the safety assessment
of many conventional food or other products that are generally regarded as safe
(GRAS).
What is Botanical Integrity? The
concept of Botanical Integrity
evolved from an initiative by the National Center for Complementary and
Integrative Health (NCCIH, formerly NCCAM) and the Office of Dietary
Supplements (ODS), both at the US National Institutes of Health (NIH). The
initiative led to the implementation of the NIH natural products integrity
policy,1 which addresses botanical study materials by outlining
special requirements for their characterization. Going beyond just “quality
control,” Botanical Integrity
combines disparate aspects of defining and assessing plant-derived materials
and products for human consumption: identity (correct plant species and plant
part), homogeneity (absence of contaminations by other species and chemicals,
often called “purity” but different than the regulatory use of that term),
biological potency (the presence of bioactive principles in desired amounts; a
prerequisite for in vivo efficacy), and safety (an adequate toxicological
profile).
How can Botanical Integrity be
assessed? The assessment of Botanical
Integrity requires a multidisciplinary approach that combines three major
domains of expertise, with each providing a variety of tools and analytical
methodologies (See Fig.
1): botanical examination (Botany),
phytochemical analysis (Chemistry),
and biological and safety assessment (Bioactivity).
Botanical examination is performed on the source material and involves
operations typically performed in plant taxonomy, morphology, and genetics.
Phytochemical analysis can be carried out with crude plant material, extracts,
and finished products. These analyses encompass a wide array of chemical
analytical tests with major domains in chromatography (e.g., LC, GC, TLC) and
spectroscopy/spectrometry (UV, (N)IR, MS, NMR). Operations for the assessment
of bioactivity and safety, which are typically performed with botanical
extracts, comprise in vitro and sometimes in vivo assays for endpoints related
to the health benefit and/or safety of the herb. Investigations in the UIC/NIH
Center for Botanical Dietary Supplements Research have integrated various
aspects of all three of these fundamental domains (botany, chemistry, and bioactivities)
since 1999. The UIC/NIH Center has pioneered new methodologies for the
assessment of Botanical Integrity and
produced insights into the safety and efficacy of botanical dietary supplements
that are widely used for women’s health. We have also implemented Botanical Integrity Dossiers as a means
of documenting comprehensively the Botanical
Integrity of our study materials, including crude herbal material, extracts,
and purified phytochemicals.
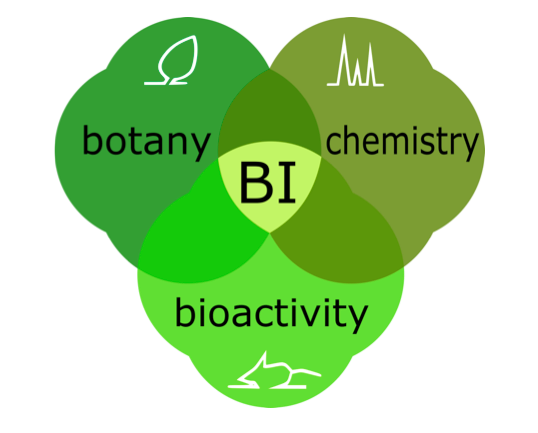
Figure 1. Botanical Integrity Requires Integration: Chemistry x Botany x
Bioactivity = Integrity. The three main components of the Botanical
Integrity (BI) model used for research in the UIC/NIH Botanical Center are:
botanical examination (botany), phytochemical
analysis (chemistry), and biological
and safety assessment (bioactivity).
The concerted use of multiple methodologies from all three components is
required to obtain a comprehensive representation of a botanical material.
How does Botanical Integrity help
ensure safety and efficacy? Under the governance of the Dietary Supplement
Health and Education Act (DSHEA) of 1994, botanical products may be marketed as
dietary supplements as long as they meet the appropriate regulatory
requirements within that category of food, not drug, regulations. Category-specific
current good manufacturing practices (cGMP) and labeling requirements are in
place for botanical dietary supplement manufacturers, and have fostered the
development of a marketplace with a broad range of high-integrity products.
However, adulteration and other issues affecting the Botanical Integrity of products can still occur, and the
combination of botanical, chemical, and biological standardization is a viable
approach for ensuring safe and effective botanical products.
What is DNA Barcoding? The authentication of botanical raw materials
(unextracted fresh and dried plant parts) has benefitted from the
implementation of a variety of molecular biology methods based on the analysis
of DNA sequences, and was originally employed for plant systematics (i.e., the
classification and naming of plants).2-4 Among these methods, DNA
barcoding relies on the amplification and sequencing of short (400-800 base
pairs) nucleotide sequences localized to standardized regions of the genome
(plastid and nuclear loci for plants). Species are identified according to the
nucleotide variations in these short standardized gene regions, which give them
unique DNA identification tags.2,3,5 During the last decade, DNA
barcoding has emerged as a method for the authentication of plant parts and
herbal materials.6-10
Recently, a DNA-based authentication method was employed to evaluate the
identities of 78 samples from commercial botanical dietary supplement products
sold in New York, allegedly to expose potential cases of fraud.11-13 The
study generated much discussion after concluding that only 21% of products
contained DNA of the claimed plant species, thereby concluding that the other
79% of the products were not in conformance with their labels. This report also
raised the question as to whether DNA barcoding and DNA-based authentication
methods are sufficient and/or conclusive for the determination of botanical
identity, and/or to what level DNA methodology addresses Botanical Integrity.
What is the Role of Plant DNA Authentication in Botanical Integrity?
DNA-based methods such as DNA barcoding are one of the many available tools
that can be used in botanical examination (“Botany,” See Fig. 1). The key outcome of
botanical examination is the positive identification of the plant or
plant-derived material by a scientifically valid method; the effectiveness of
botanical assays based solely on DNA authentication depends on the presence of
tissue and/or DNA from the target plant. In addition to DNA barcoding, there
are other useful methods of botanical examination, such as morphology with the
use of taxonomic keys, microscopy, and organoleptic analysis. In addition, a
myriad of well-established methods are available to perform phytochemical
analyses (e.g., thin-layer chromatography [(HP)TLC], liquid chromatography
[(U/HP)LC-UV, LC-MS], near infrared spectroscopy [NIR], mass spectrometry [MS,
MS/MS], and nuclear magnetic resonance [NMR]) and bioactivity assessments
(e.g., in vitro and in vivo assays for a variety of biological activities such
as estrogenicity,14 chemopreventive,15,16 and anti-inflammatory
activities.17). Importantly, chemical and biological assays can
provide strong evidence about Botanical
Integrity even when original plant tissue or DNA is absent, such as in
botanical extracts or final products containing them, or when the test
materials contain other constituents such as excipients that may confound the
analytical challenge. All three types of methodologies together (botany, chemistry, bioactivity) establish Botanical Integrity and it is incomplete
unless adequate phytochemical analyses have been performed. The complex
chemical composition of botanicals requires parallel bioanalytical approaches
for adequate description, especially when considering that the bioactive
constituents in botanicals are (phyto)chemicals.
Conclusions. The assurance of Botanical
Integrity is of paramount importance to ensure safe and efficacious
botanical-derived ingredients that can support both high-quality products and
scientific excellence. Claims made about the “quality,” or lack thereof, of
botanicals can be valid only if they rest on a variety of established and
validated analytical methods (botany, chemistry, bioactivity) that are
sufficient to address Botanical Integrity
(See Fig. 1). In our experience, the
employed methods have to be complementary to each other and cover at least two
(botany and chemistry) — if not all three — of the major domains of expertise, leading
to botanically, chemically, and biologically standardized materials.18
Notably, this process can be quite demanding, considering that several
different (orthogonal) methods should be used to cover each domain of
expertise.
In our scientific judgment, performing just one form of analysis, e.g., DNA barcoding, is insufficient to fully
assess Botanical Integrity.
Single-track approaches generally lack the rigor essential for authenticating
the identity and adequately describing the biomedical integrity of the test
material. DNA-based methods can be valuable tools for botanical evaluation in
the early stages of the botanical supply chain. However, for the analysis of
commercial botanical extracts and finished dietary supplement products, the numerous
other methods belonging to the domains of phytochemical analysis and
bioactivity assessment are much more meaningful and practical.
References
- NCCIH policy: natural
product integrity. National Center for Complementary and Integrative Health
website. Avaialble at:
https://nccih.nih.gov/research/policies/naturalproduct.htm?lang=en. Accessed
April 2, 2015.
- Hebert PDN, Gregory TR. The promise of DNA barcoding for taxonomy. Systemat Biol. 2005;54(5):852-859.
- Ajmal Ali M, Gyulai G, Hidvégi N, et al. The changing epitome of species
identification – DNA barcoding. Saudi J
Biol Sci. 2014;21(3):204-231.
- Chen S, Pang X, Song J, et al. A renaissance in herbal medicine identification:
From morphology to DNA. Biotechnology
Advances. 2014;32(7):1237-1244.
- Li M, Cao H, But P-H, Shaw P-C. Identification of herbal medicinal materials
using DNA barcodes. J Systemat Evol. 2011;49:271-283.
- Stoeckle M, Gamble C, Kirpekar R, Young G, Ahmed S, Little D. Commercial teas
highlight plant DNA barcode identification successes and obstacles. Sci Rep. 2011;1:42.
- Baker D, Stevenson D, Little D. DNA barcode identification of black cohosh
herbal dietary supplements. Journal of
AOAC International. 2012;95(4):1023-1034.
- Little DP. Authentication of Ginkgo
biloba herbal dietary supplements using DNA barcoding. Genome. 2014;57(9):513-516.
- Techen N, Parveen I, Pan Z, Khan I. DNA barcoding of medicinal plant material
for identification. Current Opinion in
Biotechnology. 2014;25:103-110.
- Kazi T, Hussain N, Bremner P, Slater A, Howard C. The application of a
DNA-based identification technique to over-the-counter herbal medicines. Fitoterapia. 2013;87:27-30.
- Study : Many Herbal Supplements Aren’t What the Label Says. The New York Times. February 3, 2015.
Available at: http://www.nytimes.com/aponline/2015/02/03/us/ap-us-herbal-supplements-investigation.html?_r=0. Accessed April
2, 2015.
- Seres DS. The potential danger of dietary supplements. CNN. February 5, 2015. http://www.cnn.com/2015/02/05/opinion/seres-herbal-supplements/. Accessed February
15, 2015.
- ABC Says New York Attorney General Misused DNA Testing for Herbal Supplements,
Should Also Have Used Other Test Methods as Controls. [press release] Austin,
TX: American Botanical Council; February 3, 2015. Available at: http://cms.herbalgram.org/press/2015/ABCSaysNYAttyMisusedDNA.html. Accessed February
10, 2015.
- Hajirahimkhan A, Simmler C, Yuan Y, et al. Evaluation of estrogenic activity of
licorice species in comparison with hops used in botanicals for menopausal
symptoms. PLoS ONE. 2013;8(7):e67947.
- Dietz B, Liu D, Hagos GK, et al. Angelica sinensis and its alkylphthalides induce
the detoxification enzyme NAD(P)H:quinone oxidoreductase 1 by alkylating KEAP1.
Chem Res
Toxicol. 2008;21:1939-1948.
- Dietz B, Hagos GK, Eskra JN, et al. Differential
regulation of detoxification enzymes in hepatic and mammary tissue by hops (Humulus lupulus) in vitro and in vivo. Mol Nutr Food Res. 2013;57(6):1055–1066.
- Hajirahimkhan A, Dietz BM, Bolton JL. Botanical Modulation of Menopausal
Symptoms: Mechanisms of Action? Planta
Medica. 2013;79(7):538-553.
- Farnsworth NR, Krause EC, Bolton JL, Pauli GFF, van Breemen RB, Graham JG. The
University of Illinois at Chicago/National Institutes of Health Center for
Botanical Dietary Supplements Research for Women's Health: from plant to
clinical use. Am J Clin Nutr. 2008;87(2):504S-508S.
|