As is well known to many people in the medicinal plant research
community and dietary supplement industry, dietary supplements, including
herbal products, are not subject to the same regulatory guidelines for
pre-market testing as conventional pharmaceutical drugs in the United States. This
is due to a variety of regulatory reasons regarding dietary supplements’ being
viewed more as foods rather than drugs that are not relevant for further
discussion here. However, some health professionals and others have expressed
varying degrees of interest and concern regarding the potential for clinically
relevant interactions between conventional pharmaceutical medications and
herbal and other dietary supplements.1,2 Considerations such as
history of safe use (within context of traditional versus modern usages),
literature data from pharmacology and toxicity studies, and constituent amounts
in supplement products provide some guidance on whether to assess herb-drug
interactions (HDIs) experimentally. The scientific literature is replete with pre-clinical
reports of various herbal extracts and constituents as potent inhibitors of
drug-metabolizing enzymes (Table 1).3-6 However, without the use of appropriate analytical methods for
herbal product characterization and quantitation of constituents, dose
performance analysis, or in vitro
testing in physiologically relevant models to allow some prediction of
bioavailability of key constituents, extrapolating these reports to determine
whether human testing is necessary to identify clinically relevant HDIs is
difficult. This lack of a clear determination of risk hinders clinicians and
consumers from making informed decisions about the safety of taking herbal
products with conventional medications. A suitable framework is needed that
describes a flexible approach for assessing when human HDI studies are
warranted, and an outline of standard methods when HDI testing is conducted.
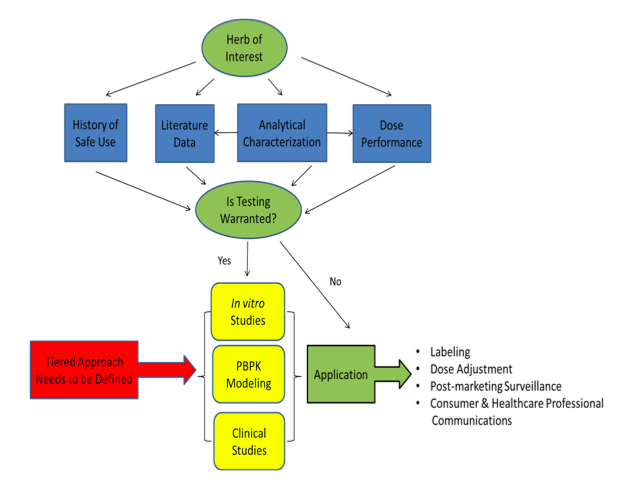
Figure
1. A summary of the key components that should be included in a common
framework for assessing potential herb-drug interactions (HDIs).
Herbal product usage in Western countries continues to increase across
all age groups.7,8 Individuals in these countries also have ready
access to conventional medications, and significant polypharmacy is often
observed, particularly in women and older adults.8,9 Many patients
are reticent to disclose herbal product usage to their healthcare providers,
and many providers still do not inquire about such usage. In addition, many
healthcare professionals are now recommending herbal products to counteract
side effects of some conventional drugs.10 Thus, the potential for
both HDIs and drug-drug interactions is frequently ignored in clinical practice
because of the complexity of the problem. The net result is that the
opportunity for clinically significant HDIs
exists and should be evaluated in a systematic manner.
Although dietary supplements and herbal products in most countries are
not subject to the same regulatory guidelines for pre-market testing as conventional
drugs, there is an increasing focus by many regulatory agencies on the
potential for HDIs. Likewise, consumers have an increased awareness of HDIs as
a result of numerous media reports. Providing accurate information on potential
HDIs facilitates informed decision-making by consumers and healthcare
providers.
Table 1. Examples
of potent in vitro CYP450 and/or
transporter inhibition by herbal extracts
or constituents as reported in the scientific literature with no
current
follow-up as to clinical relevance.
|
Herb and plant part / Individual Constituent(s)*
|
CYP450/Transporter Inhibited
|
Reference
|
|
Frankincense tree resin extract, (Boswellia serrata, Burseraceae)
|
CYP1A2, 2C8, 2C9, 2C19, 2D6, 3A4
|
3
|
|
β-Boswellic acid, 11-keto- β-Boswellic
acid
|
CYP2C8, 2C9, 3A4
|
3
|
|
Acetyl-11-keto- β-Boswellic
acid,
|
CYP2C9, 3A4
|
3
|
|
Acetyl- β-Boswellic
acid
|
CYP2C9
|
3
|
|
Schisandra fruit (aka five-flavor-fruit) extract,
(Schisandra chinensis, Schisandraceae)
|
CYP3A4
|
4
|
|
Gomisin C, Gomisin B, Gomisin G
|
CYP3A4
|
4
|
|
Gomisin N, Gomisin A
|
CYP2C19, 3A4
|
4
|
|
Rhodiola root (aka Golden root) extract, (Rhodiola rosea, Crassulaceae)
|
CYP3A4, P-gP
|
5
|
|
Cat’s claw herb extract, (Uncaria tomentosa, Rubiaceae)
|
CYP3A4
|
6
|
* Standardized common name and Latin binomial,
family name per American Herbal Products
Association’s Herbs of Commerce, 2d ed. (2000),14 and United
States Department of Agriculture’s Agriculture Research Service (ARS) GRIN
database.15
There are an increasing number of scientific papers related to the field
of HDIs. However, most studies utilize simple in vitro metabolic systems (e.g., liver microsomes), and the
results are likely too unreliable to provide meaningful assessment of
clinically relevant HDI potential. The exposure in vitro to the complete phytochemical complexity of an herb or
herbal extract does not represent the systemic exposure to the ingested and
altered phytochemical matrix or its limited absorption and variable
distribution. Thus, most of these reports are preliminary and often do not
attempt to define the clinical relevance of such findings. There is little
follow-up work conducted in more complex in
vitro systems such as whole-cell hepatocytes with fully functional
transporter and metabolizing enzymes, or the use of physiologic-based
pharmacokinetic (PBPK) models to
extrapolate to in vivo relevance. Won
et al. recently reviewed a number of dietary substance-drug interactions in
which both in vitro and clinical data
exist, and in many cases there was no correlation of findings.11 In
addition, there is sometimes poor analytical characterization of the botanical materials
used, contributing in large part to inconsistent findings across studies.
In the prescription medicine world, there is clear guidance on how to
assess potential drug-drug interactions.12,13 Because there is no
standard/systematic regulatory guidance on testing for HDI potential, there is
an opportunity in the scientific community to lead the way in establishing a
framework for assessing HDI potential.
From a dietary supplement industry perspective, an ideal framework approach
for assessing HDI potential would include the following criteria:
- A
screening approach that can encompass a pipeline (i.e., “higher throughput”);
- Cost
and resource efficiency;
- Must be
readily transferable to external partners (e.g., contract research
organizations) since many companies do not have the internal expertise and/or
testing facilities;
- Be
consistently applied across the industry;
- Include
a decision tree on when more in-depth studies may be warranted (e.g., tiered
approach);
- Provide
guidance on how to design and interpret studies;
- Provide
guidance on how to apply HDI information to dose adjustment, labeling, and/or
post-marketing surveillance strategy.
The key components that should be included in a common framework for
assessing HDI potential are summarized in Figure 1. Likewise, there are a
number of important considerations that should be included within each
component (Table 2). Follow-up studies may be warranted in situations where
there is an inconsistent history of safe use or insufficient literature data on
HDI potential — e.g., no data available on cytochrome P450 (CYP450)/transporter
inhibition/induction potential, or literature data reporting potent in vitro inhibition of
CYP450s/transporters. When testing is necessary, one can borrow, where
relevant, from the drug-drug interaction guidances of the US Food and Drug Administration
and European Medicines Agency.12,13
Table 2. Important considerations that warrant inclusion within each
component of a framework for assessing potential HDIs.
|
History of Safe Use:
- How do geography and culture of historical use compare to
proposed product market?
- Is historical use the same as proposed product use?
- Same form (whole plant vs. plant part vs. single ingredient)?
- What is known about the consumer population that product targets
(acute vs. chronic use, underlying disease/conditions, co-medications,
age group)?
|
|
Literature Data:
- PK studies on constituents provide understanding of which
constituents are readily absorbed and what relevant concentrations to
use in in vitro assays.
- Which drug metabolizing enzymes/transporters are affected may
guide the need to do additional studies (e.g., potent inhibition of
CYP3A4 would likely be more concerning than moderate inhibition of
CYP1A2).
- Are there clues in the clinical chemistry and/or histopathology
from animal toxicity studies that may indicate potential effects on drug
metabolizing enzymes or transporters (e.g., increases in bilirubin,
cholestasis, increased liver weight, etc.)?
|
|
Incorporation of Analytical
Characterization:
- Useful for assessing toxicity potential, but can also be applied
to assessing HDI potential.
- Enables further data mining of literature for HDI information.
- Are there any structure-activity relationship (SAR) alerts for
individual constituents of the herbal extract/constituent?
- Quantitation of individual constituents can be useful in
predicting potential exposure levels, designing in vitro studies, or whether additional testing is necessary
(cost effective).
|
|
Dose Performance:
- Disintegration of dose form
- Dissolution of constituents
- Physical-chemical data on constituents
- Solubility information on extract/constituents
|
In summary, there is a need to form an
academia/industry/regulatory-wide expert working group to develop a framework
for assessing HDI potential. Expertise is needed in diverse areas including in vitro metabolism/transporter studies,
PBPK modeling, clinical pharmacokinetics, analytical chemistry,
biopharmaceutics, and risk assessment. The objective of an expert working group
would be to develop a comprehensive strategy that incorporates these key
components into an overall HDI “risk assessment” that facilitates informed
decision-making by consumers and healthcare providers.
The topic of assessing HDI potential will be highlighted at the 41st
Annual Summer Meeting of the Toxicology Forum, July 12-16, 2015. A plenary
presentation entitled, “Assessing Potential Natural Product-Drug Interactions: Need
for a Common Framework Approach,” which further defines the concepts captured
herein, will be presented by a number of experts in this field. Details related
to this meeting can be found at: http://toxforum.org/next_meeting.
Amy L. Roe, PhD, is a senior toxicologist in the personal
healthcare division at The Procter & Gamble Company in Cincinnati, Ohio.
She received her PhD in toxicology from the University of Kentucky in 1997 and
conducted post-doctoral work at the University of Cincinnati. Her expertise
includes general and regulatory toxicology, drug/xenobiotic metabolism, and
pharmacokinetics. She is a diplomate and current board member of the American
Board of Toxicology, and serves as councilor on the Regulatory and Safety
Evaluation Specialty Section of the Society of Toxicology. She can be contacted
at roe.al@pg.com.
Disclosure
The Procter & Gamble Company is a distributor of dietary supplement
products.
Acknowledgements
The author would like to acknowledge discussions and input to this
framework approach from Mary Paine, PhD (Washington State University), Bill
Gurley, PhD (University of Arkansas for Medical Sciences), Rick Kingston, PharmD
(SafetyCall International), Hellen Oketch, PhD (United States Pharmacopeia), and James Griffiths, PhD (Council
for Responsible Nutrition).
References
1. Melville
N. ‘Polyherbacy’ a common challenge in pain patients. Medscape. September 23, 2014. Available at: www.medscape.com/viewarticle/832191.
Accessed April 27, 2015.
2. US Food
and Drug Administration. Mixing medications and dietary supplements can endanger
your health. FDA Consumer Health
Information. October 2014:1-2. Available at: www.fda.gov/ForConsumers/ConsumerUpdates/ucm420349.htm.
Accessed April 27, 2015.
3. Frank A,
Unger M. Analysis of frankincense from various Boswellia species with inhibitory activity on human drug metabolising
cytochrome P450 enzymes using liquid chromatography mass spectrometry after
automated on-line extraction. J Chromatogr
A. 2006;1112(1-2):255-262.
4. Iwata
H, Tezuka Y, Kadota S, Hiratsuka A, Watabe T. Identification and
characterization of potent CYP3A4 inhibitors in schisandra fruit extract. Drug Metab Dispos. 2004;32(12):1351-1358.
5. Hellum
BH, Tosse A, Hoybakk K, Thomsen M, Rohloff J, Nilsen OG. Potent in vitro inhibition of CYP3A4 and
P-glycoprotein by Rhodiola rosea. Planta Med. 2010;76(4):331-338.
6. Budzinski
JW, Foster BC, Vandenhoek S, Arnason JT. An in
vitro evaluation of human cytochrome P450 3A4 inhibition by selected
commercial herbal extracts and tinctures. Phytomedicine.
2000;7(4):273-282.
7. Gahche
J, Bailey R, Burt V, et al. Dietary supplement use among U.S. adults has
increased since NHANES III (1988-1994). NCHS
Data Brief. 2011;(61):1-8.
8. Djuv A,
Nilsen OG, Steinsbekk A. The co-use of conventional drugs and herbs among
patients in Norwegian general practice: a cross-sectional study. BMC Complement Altern Med. 2013;13:295. doi:
10.1186/1472-6882-13-295.
9. Farina
EK, Austin KG, Lieberman HR. Concomitant dietary supplement and prescription
medication use is prevalent among US adults with doctor-informed medical
conditions. J Acad Nutr Diet. 2014;114(11):1784-1790.e2.
Available at: www.andjrnl.org/article/S2212-2672(14)00106-3/pdf. Accessed
April 27, 2015.
10.Reddy
S. A top hospital opens up to Chinese herbs as medicines: Evidence is lacking
that herbs are effective. The Wall Street Journal. April 21, 2014. Available
at: http://www.wsj.com/articles/SB10001424052702303626804579509590048257648.
Accessed April 27, 2015.
11.Won CS,
Oberlies NH, Paine MF. Mechanisms underlying food-drug interactions: inhibition
of intestinal metabolism and transport. Pharmacol
Ther. 2012;136(2):186-201.
12.U.S. Department
of Health and Human Services, Food and Drug Administration, Center for Drug
Evaluation and Research (CDER). Guidance for industry: drug interaction studies
— study design, data analysis, implications for dosing, and labeling
recommendations. February 2012:1-79. Available at: www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm292362.pdf. Accessed
April 27, 2015.
13.European
Medicines Agency. Guideline on the investigation of drug interactions. June 21,
2012;1-60. Available at: www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2012/07/WC500129606.pdf. Accessed April 27, 2015.
14.McGuffin
M, Kartesz JT, Leung AY, Tucker AO, eds. American
Herbal Products Association’s Herbs of Commerce, 2nd ed. Silver Spring, MD:
American Herbal Products Association; 2000.
15.USDA,
ARS, National Genetic Resources Program. Germplasm Resources Information
Network (GRIN) Online Database, National Plant Germplasm Sytem. Available at: www.ars-grin.gov/npgs/. Accessed April
27, 2015.
|