Editor’s Note: The following article is a supplement to the HerbalGram article, "Lunar Influence: Understanding Chemical Variation and Seasonal Impacts on Botanicals," written by Ian B. Cole and Michael J. Balick, PhD, and published in 2010 in HG issue 85. It was originally published in Aux Origines des Plantes – Des plantes anciennes à la botanique du XXIè siècle. (F. Hallé éd.). Fayard, Paris., p. 389 – 411, and has been slightly edited for publication by ABC.
By Ernst Zürcher, PhD
A Parallel
When the annual reunion of the Swiss Natural Sciences Society was held in Lucerne in 1834, the German geologist Jean de Charpentier—a defender of the modern theory of glaciations— was able to attend and present the results of his work. As he journeyed to this learned occasion, he fell into conversation near the Brünig pass with a woodcutter from Meiringen. When the latter saw the stranger examine a block of granite lying near the path, he said to him, “There are many stones like this one around here, but they come from far off, from the Grimsel mountains; for it's Geissberger [a kind of rock], and the mountains here aren't Geissberger.” When asked how these stones could have reached this spot, the mountain dweller replied without hesitation: “It's the Grimsel glacier which brought them here, for at one time it extended as far as the town of Berne; there's no way the water could have dropped them here, so far from the bottom of the valley.” What an encounter! The famous professor, with his lecture about the theory of glaciations in his briefcase, hears an anonymous woodcutter expound as the most obvious facts, matters which the scientists would argue about for years more before they became part of the world scientific heritage.
Tree Growth and Properties of Wood: Rhythms and Traditions
Most organic processes—and the structures that result from them—present a rhythmic character. Thus, trees have a characteristic rhythmic growth with phases of activity and of rest. This form of growth occurs not only when the environment provides seasonal variations, but also when conditions are practically constant, such as is the case in equatorial regions, for example. Rhythmic variations can be observed at practically all development stages, from germination to seed formation. This systematic alternation between activity and rest phases can also be seen in the morphology of shoots or the crown architecture and, at an anatomical level, in the succession and internal structure of growth rings.
Here the question arises about the cause and nature of these rhythmic phenomena: is it simply an interaction between the effect of the sun on a daily level and of the seasons and “mechanisms” endogenous to the organism? Could it not also involve other types of connections of plants with celestial movements, in particular, those of the moon?
In order to survive, humans—being heterotrophs (using the substance of other organisms as food)—have always had to take into account this rhythmic character of plants. On perusing works about folk customs and country lore, or reading the accounts of ancient authors (such as Hesiod, an 8th century B.C. Greek poet, author of Works and Days), or again when talking with gardeners, small farmers, woodcutters, or those working with wood about their empirical experience, one is led unerringly to 2 observations:
• In addition to the rhythm of the seasons, solar in nature from a geocentric point of view, these sources and these people systematically evoke the lunar cycles as a factor influencing the growth, the structures and certain properties of plants;
• Despite the geographical distance between the informants, there are often similarities among the declarations concerning this factor.
This would seem to indicate the possible existence of real phenomena. Thus, general maxims about felling trees agree for they mention the “moon” factor in the Alps, the Middle East, in India, Ceylon or Brazil, in Guyana, Korea or Finland. All these traditions seem to be based on similar observations, for example, the period of the new moon (or waning moon) is considered the most favorable for felling trees to give durable wood for construction, resistant to insects and fungi. A “phytopractice” of this kind concerns the period for planting cuttings in Central America, considered optimal in a waning moon.
It should be pointed out that in former times, people could observe for longer periods of time and under calmer conditions. Observation must even have been of vital importance to them, since survival was more precarious than now. Until recently, no machine, no process of automation came between humans and the object of their work, which probably allowed them to refine their perception to a considerable degree.
A portion of superstition probably added itself, as soon as precise and objective observation gave way to the blind acceptance of traditional maxims. The change from oral to written transmission probably added its share of distortions.
3 Types of Forestry Maxims
For the determination of the “right felling date,” historical records and oral tradition provide 3 types of maxims:
• According to the “phases of the moon,” the synodic lunar rhythm has a period of 29.531 days. It measures the passage of a new moon to a full moon (“waxing” phase), and on to the next new moon (“waning” phase; see Figure 1). On a daily level, this synodic rhythm may be observed in the movement of the tides, the principal period of which is 24.8 hours, but this is rarely mentioned in tradition; the only case presently known to the author concerns the cutting of bamboo ( Phyllostachys aurea) in Ecuador: in order to determine the times of low tide that are favorable to the conservation of bamboo, the inland farmers cut a certain “water liana,” which, at these particular moments, is apparently devoid of exudation. 2 Figure 1: Phases of the Moon (corresponding to the synodic rhythm, with a revolution in 29.5 days) 1) Lunar orbit arount the Earth, for an observer looking at the Earth-Moon system from a long way above the Earth's North Pole 2) New Moon, observed from the Earth 3) Waxing crescent 4) First quarter 5) Full Moon 6) Last quarter 7) Waning crescent 8) The Earth 9) Direction of solar rays. Between the first quarter and the full Moon, the phase is called "waxing gibbous;" between the full Moon and the last quarter is "waning gibbous." • According to the tropical lunar rhythm, the cycle of the “ascending moon” and the “descending moon” is relative to the terrestrial horizon. This rhythm, less obvious to the observer, concerns the height of the lunar trajectory relative to the horizon, which varies systematically. The height of the trajectory increases 13 or 14 times, then decreases over the second half of the tropical month, which lasts exactly 27.32158 days. The tropical and synodic cycles should not be confused, for their cycle length differs by 2.21 days. These 2 rhythms always coincide toward the end of December, with the full moon and the new moon occurring respectively at the highest and lowest points of the tropical trajectory.
• According to the constellations, or signs of the Zodiac, in which the moon is found, there is a third level of influence, frequently mentioned through the ages; some documents, such as the Manichean writings, go back to Persian times.3 This is the sidereal lunar rhythm, which has a period very similar to the tropical rhythm, and is based on the constellations of the Zodiac, in front of which the moon passes during its orbit around the Earth. The cycle is completed in 27.32166 days. Error may be caused over the long term by the fact that the astronomical constellations observed at a given date vary infinitesimally because of the slow gyration (precession) of the Earth's axis.
Practices That Remain Very Much Alive
Today still, maxims about felling linked to the moon are applied by certain workers of wood. Here, we will not deal with the multiple “lunar calendars”—very trendy nowadays—covering numerous areas fairly comprehensively without any experimental basis. The examples that follow concern cases known directly to the author, or are taken from scientifically documented sources; their aim is to illustrate the great variety of uses of wood for which the moon factor is considered important for obtaining certain exceptional properties. It should be pointed out that in most cases this factor comes only in second or third position, as the most important factors are the general time of year, with a high value placed on “winter wood,” and growing conditions, with mountain wood from slow-growing natural forest stands being particularly appreciated. Sometimes winds are mentioned, such as the Foehn in the Alps, which could negatively impact certain properties of the wood.
Wood for construction
The principal properties required here are mechanical resistance (to pressure, traction, or flexion) and resistance to fungal and insect attack. A French maxim stipulates, Bois tendre en cours / Bois dur en décours, or “soft wood in a waxing moon, hard wood in a waning moon.”
Wood shingles
These are boards of different dimensions used as tiles for covering roofs and façades, hence are particularly exposed to the weather and rotting. Appropriate types of wood can come only from well-proven species, such as oak (Quercus spp.), sweet chestnut (Castanea sativa) or larch (Larix spp.). There are also spruce (Picea abies) and fir (Abies spp.), that some shingle-makers fell at particular lunar phases in order to obtain a material that dries rapidly after each shower of rain.
Chimneys
In certain parts of Central Europe, wood was used even in chimney construction, and also used to smoke meat. Even for these uses, there are “lunar” felling maxims that are supposed to provide wood that is not easily inflammable.
Firewood
Still commonly applied in the Jura, for example, the maxim mentioned by Olivier de Serres advises: “the position of the moon is noteworthy: when waxing, cut firewood, when waning, cut wood for building.”4 In this case, it is clearly combustibility that is sought, rather than hardness or resistance to destructive agents.
Resonance wood
Right up to the most prestigious use, as the “sounding board” in making violins, guitars, or pianos, the date of felling relative to the moon's phases often constitutes one of the secrets of the instrument-maker's art.
Barrels and casks
According to the observations of craftsmen, the most watertight oaken staves cannot be obtained at just any season of the year, nor at just any phase of the moon.
Bamboo
Traditions widespread in South America (Columbia, Ecuador, Brazil), and also in India, take into account the lunar calendar for cutting bamboos (which are in fact giant Gramineae or grasses) capable of resisting insect attacks.
Floating
Several independent traditional sources mention that, not only the properties of wood, but also the way the lumber floats on rivers will vary according to the periods of the moon.
Challenges for Research
In the face of all these traditions and practices stemming from the world of forestry (agriculture could provide similar ones), initially surprising given modern knowledge, the scientific world finds itself questioning. The researcher finds before him or her, the challenge of examining objectively and critically whether there might not be an inkling of truth in this “ancient wisdom,” whether one or several real phenomena or processes might not lie behind this craft lore. The facts must be differentiated from the superstitions, and then quantified; the researcher must be prepared to question and enlarge some of our present theories. If certain phenomena were to be confirmed, even partially, we would find ourselves enriched by a great treasure born of the centuries-long contact of humans with nature, offering us new work hypotheses and development possibilities. The situation is analogous to that of pharmaceutical research, guided by the insights of traditional pharmacopeia, whose specificity and efficacy are often astonishing.
This approach has already been undertaken a number of times in the framework of modern scientific methodology for annual plants; sometimes, researchers found themselves unexpectedly faced with lunar rhythms. The potential for innovation still needs to be communicated to the scientific community and to specific users.
Chrono Terms
The discipline studying the temporal structures of organisms is called chronobiology. It examines all the rhythmical manifestations of vital processes—meaning, in addition to lunar rhythms, daily and yearly rhythms, those around the hour or the minute (for example, the human digestive system), or those of even higher frequency (around a millisecond, such as those operating in the nervous system). Chronobiology has developed considerably over the last 50 years, with specializations in different directions. Chronophysiology describes and analyzes the configuration of the rhythmic processes of different functions of the organism and of their interactions with environmental factors. Chronopharmacology analyses the chronobiological effects of medicines. Modern medicine includes chronopathology, chronotoxicology, chronotherapy, and chronohygiene.
The principal lunar cycle, the synodic cycle, can be divided into sub-cycles linked to the phenomenon of the tides, due to the daily rotation of the Earth. The following can be distinguished:
Period Rhythm or Lunar Cycle Unit
12.42 hours Tidal or lunitidal Twice-daily tide
[24.0 hours Daily Day: solar cycle, reference]
24.85 hours Tidal day, lunidian Lunar day
7.38 days Circaseptan Lunar week
14.76 days Semi-lunar Syzygy (conjunction or opposition in relation to the sun)
29.53 days Lunar, synodic Lunation, lunar month
Lunar Rhythms in Biology: An Overview
Endres and Schad (1997) put forward the reality of lunar periods in the annual plant world, in animal biology, and in humans.5 A series of more than 600 species—mostly animal—showing this type of rhythm, being experimentally documented, and published in scientific journals, illustrates the complexity of the chronobiological phenomena involved, and raises the question of the fundamental nature of time. Engelmann (2004) recommends in Rhythms of Life6 the works by Pearse (1990) and by Palmer (1995) as good references on the rhythms linked to tides of 2 weeks and one month.7,8
While no lunar periodic rhythm has been reported for Cyanophyceae, (the former class of blue-green algae, presently classified as Cyanobacteria), probably due to their life cycle that is short relative to the lunar rhythms, 45 species of Thallophytes and 40 species of Angiosperms were already listed in 1996.
All eukaryotic cells and organisms oscillate in general between exogenous and endogenous factors, due to their internal clock based on interactions between the nucleus and the cytoplasm. In 1938, Bracher discovered that in the mud of the West coast of England, the protist Euglena limosa migrates not only with a circadian (daily) rhythm, but also according to a lunar rhythm (24.8 hours), which allows it to appear at the surface only by day and at low tide.9 This unicellular species with a distinct nucleus has movement through its flagella, chlorophyllic assimilation, and a rudimentary eye. This demonstrates that the lunar rhythmic factor, like the solar factor (photoperiodic), constitutes a continuum including many living forms, both plants and animals.
Lunar Periodicities in the Germination, Growth, and Fructification of Annual Plants
One of the first research projects run according to scientific criteria was by Semmens and published in 1923,10 with precisions added in 1947, in Nature.11 The researcher mentions the germination of the seeds of mustard (Sinapis spp.) strongly accelerated by the effect of moonlight. She establishes experimentally a relation between this phenomenon and the polarized nature of this light due to its reflection, a polarization that is more pronounced in certain moments of the cycle. This type of light accentuates the process of decomposition of the grains of starch by diastase, an enzyme mobilizing by hydrolysis this storage substance.
Through extremely exhaustive laboratory trials lasting from 1927 to 1935, Kolisko demonstrated the variations in growth of plants according to the lunar synodic rhythm. The seeds of vegetable, floral, and cereal species (lettuce [Lactuca sativa], white cabbage [Brassica oleracea], leek [Allium porrum], tomato [Lycopersicon esculentum], pea [Pisum sativum], common bean [Phaseolus vulgaris], lovage [Levisticum officinale], yarrow [Achillea millefolium], lemon balm [Melissa officinalis], monkshood [Aconitum napellus], corn [Zea mays], wheat [Triticum spp.], oats [Avena sativa], barley [Hordeum vulgare]) sown 2 days before the full moon showed better germination and stronger growth, formed more numerous inflorescences, and produced a better harvest than the seeds sown 2 days before the new moon. Some experimental series showed an abrupt change between the ideal period, about 2 days before the full moon, and the exact day of the full moon, which reveals a form of phase difference between the visible course of our satellite and its effect on germination.12 Kolisko worked with very homogeneous material and followed for the choice of dates the suggestions of Rudolf Steiner, founder of the bio-dynamic agriculture method, practiced with increasing success for more than 80 years.13
It had become evident that the exact moon phase at the moment of sowing plays an important role for the whole of the vegetative cycle that follows: germination, growth, flowering, and fruiting. This role cumulates with that of the season of the year, corresponding to the earth-sun relation.
These periodicities were subsequently confirmed, but also differentiated several times, for example, by Voegele in 1930, with several cereals tested outside, where the maximal difference appeared between the sowings 2 to 5 days before the full moon and those 2 to 3 days after the full moon.14
Popp (1933) took up Kolisko's work with different varieties of potato (Solanum tuberosum), bean, corn, and shallot (Allium ascalonicum).15 The potato showed higher yields in the batches planted towards the last quarter and the new moon, while the shallots gave their highest yield in the batches from the first and last quarter. Despite these results, the author of these trials declared that it was extremely improbable that the moon had any influence, while admitting methodological flaws, all of which provides a glimpse of the role of a priori and preconceived ideas in research touching on this theme. In a subsequent experiment, Popp (1936, 1937) again obtained marked differences for corn,16,17 but this time in the opposite way from those found by Kolisko.
In contrast, Opitz (1936) obtained higher yields from barley sown in a waxing moon than from those sown in a waning moon, with dates only one day apart.18 An analogous result is mentioned by Breda in 1960 for rye (Secale cereale), where an analysis by moving averages reveals higher yields for full moon sowings than for those at new moon.19
The systematic long-term trials undertaken by Maria Thun allow, she claims, the classification of the stimulation or the growth style of plants in 4 different types (root, stem, leaf, flower-fruit), according to the position of the moon in the Zodiac (sidereal cycle) at sowing. The results, which were unfortunately not published according to present-day scientific standards, led this researcher to edit an annual Sowing Calendar. In the 1970's Abele attests, with carrot (Daucus carota), summer barley, oats, and radish (Raphanus sativus), to the validity of Maria Thun's concepts and partially confirms the differentiation mentioned above.20,21
Spiess, using trials lasting 5 and sometimes 6 years, later provides statistical confirmation of lunar variations in the case of cultivated annuals, winter rye, radish, carrot, potato, bean, and mustard.22 In addition, Spiess highlights some interesting specificities:
• A rhythm little mentioned in traditional lore also plays a role, linked to the anomalistic revolution with a period of 27.6 days, due to the elliptical and off-center shape of the lunar trajectory. Thus, the moon passes alternately and at variable speeds near to the Earth—the perigee—then at a maximal distance—the apogee. For all the species tested, the sowings during the period of perigee show a significantly positive reaction compared to the other periods of sowing, once the effect of the season (annual trend) has been eliminated mathematically.
• An influence of the synodic rhythm (lunar phases) appears for all the species studied, with a marked difference between the waxing and waning phases. The direction of this difference is, however, species-specific: while the carrot gives maximum yield for sowings 3 days before the full moon, the yield is minimal for potato planted between the first quarter and the full moon. This confirms the results of Kolisko with the carrot, and those of Popp with the potato.23
• The tropic rhythm (ascending and descending moon) only seems to affect certain species: the common bean is the most sensitive (with a maximum number of pods for sowing with the moon at culmination—in the constellation of Gemini), followed by radish and carrot.
• Clear yield differences may be observed according to the sidereal rhythm, depending on the moon's position in the 12 Zodiac signs. Spiess points out, however, that these differences could also be explained by the first 3 rhythms mentioned; Thun's grouping into 4 types of growth according to the Zodiac cannot be confirmed by this methodology. This analysis would be made subsequently, with a positive result, by Kollerstrom and Staudenmaier (2001) reinterpreting Spiess's data.24
Similarly, Fritz's trials show radish germinating more quickly when sown 2 days before the full moon (synodic rhythm).25 By using transparent or opaque sheets of glass over the seedbeds, the author shows statistically the effect of the light of the moon on the development of leaf surface or on the stem/root ratio.
In a thesis that went practically unnoticed in 1974, Milton illustrates for corn the complexity of the synodic lunar variations by measuring the development of the young shoot (coleoptile) one week after sowing.26 Series of daily sowings in laboratory conditions indicate a weekly rhythm, by moon quarters, within which the maximum found before the full moon contrasts on the one hand with the minimum found between the third quarter and the new moon, but also with the minima of the exact moment of the first quarter and of the full moon (see Figure 2). These systematic short-period variations could explain the apparent divergence between the results of Kolisko and those of Popp for corn.
A few years before, the French scientist B. Millet had demonstrated the complexity of the elongation rhythms of the broad bean (Vicia faba): at least 5 rhythms of different periods can be identified simultaneously.27
Also of note are the rhythmic lunar variations on a monthly scale in annuals, detected by Graviou (1977), in the growth in length of the roots of cress (Lepidium sativum) kept in constant temperature and darkness, over the first 72 hours after germination.28
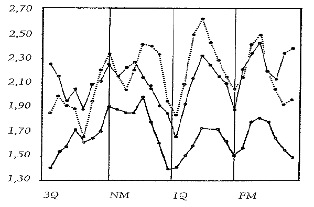 Figure 2: Development of the coleoptiles of Maize (Zea mays) in cm, 7 days after sowing, for 3 different experimental configurations.
NM=New Moon/FM=Full Moon/1Q, 3Q = 1st & 3rd quarter. Period of the trial: December 1972 to January 1974 (W.J. Milton 1974).
Germination and Initial Growth in Trees
A tree nursery situated in the tropics of Rwanda offered interesting conditions for experiments on the germination and initial growth of woody species according to the moon factor, since temperatures and day-lengths were less variable than in higher latitudes, and the dry seasons could be compensated by watering. The work occurred over 3 years: preliminary trial, main trial with 12 sowings of 4 repetitions of 50 seeds, and, lastly, the control and complementary trial. The sowings of the main trial were carried out 2 days before the full moon, alternating with sowings 2 days before the new moon, as suggested by Kolisko's work mentioned above.29
For African musizi (Maesopsis eminii) found from Liberia to Kenya, the speed and rate of germination and the mean and maximum values of growth in the first months, vary in a clearly rhythmic manner, with superior values for sowings just before the full moon (Zürcher 1992).
An independent trial carried out 5 years later in West Africa using the same method on 4 dryland species: Sclerocarya birrea, Adansonia digitata, Afzelia Africana, and Detarium microcarpum, confirms this rhythmic character, but essentially for the development stage 2 months after sowing.30
It is important in this type of trial to choose the date with great precision, for near the favorable moments between the first quarter and the full moon, the exact moment of the full moon gives results sometimes lower than those of 2 days before the new moon for musizi, which agrees with Milton's observations for corn. It is important, however, to avoid oversimplifying this relation between plant rhythms and lunar cycles. Indeed, certain species can show the opposite behavior and respond positively to a sowing before the new moon, such as blackwood wattle (Acacia melanoxylon) and sesban/Egyptian pea (Sesbania sesban), a behavior that brings to mind that of the potato observed by Popp, and later by Spiess. These first positive results concerning forest species demonstrate, nonetheless, the importance that lunar chronobiology may have for operating nurseries seeking to produce vigorous trees.
A Trial Revisited
In his short review of research on lunar rhythms in the plant world, Beeson (1946) bases his generally skeptical opinion31 on the work of Rohmeder (1938), carried out with a visibly very critical prejudice.32 This work aimed to test the phenomena recently revealed by Kolisko. The experiments were carried out meticulously with a stock of spruce seeds having a high germination rate and uniform quality, during 1936 (4 months) and 1937 (7 months), consisting of 87 series of 1,200 seeds, or a total of 104,400 seeds, whose speed and rate of germination were determined after 7, 10, 14, and 21 days. Eight dates per synodic lunar month were chosen for sowing: 1st quarter, full moon, last quarter, new moon, plus waxing crescent, waxing gibbous, waning gibbous and waning crescent (see Fig. 1). The variations having occurred each year within a relatively narrow range, the effect of seed ageing during the long series in 1937 having been stronger than the “moon” effect and the curves representing germination rate relative to lunar sowing date having presented sometimes divergent paths, all this brought the author, without any statistical analysis to the definitive conclusion that these trials “provide no argument indicating that the change in lunar phases might influence the germination of Spruce seeds.”
If we apply today, 73 years later, a statistical analysis of variance for the cumulative data from the 2 years of trials, we obtain for the variations in germination rate about its general mean a range which is indeed narrow, but comprising clearly significant differences (see Figure 3).33 These differences bring us back to the initial question about Kolisko's results, since among others, the sowings from before the full moon now appear to give significantly higher results (+2.8%) than those from before the new moon, and in addition, significantly higher than those from the exact day of the full moon, the waxing crescent and the first quarter.
Kolisko's results here find a belated confirmation for a first-order forestry species, similar to those concerning the musizi, where an abrupt fall in initial growth for sowings on the day of the full moon was observed.
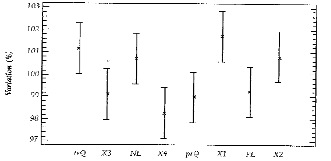 Figure 3: Variation around the general mean germination rate of Spruce at 21 days, for sowings according to moon phases (1936, 1937), re-analysed, from E. Rohmeder.1 The sowing X1, 3 days before the Full Moon, gives significantly higher results than X3, 3 days before the New Moon, and than the sowings of Full Moon (PL), First Quarter (prQ) and 3 days before (X4).
Lunar Rhythms in Cytology, Physiology, and Morphology
The variations in the germination, growth, and fructification of plants in relation to lunar cycles have their source in, or are accompanied by, a number of interesting phenomena, listed here first for herbaceous plants as promising leads for research.
Herbaceous Plants
• Analyses of DNA and mitotic activity of potato tissue cultivated in laboratory reveal variations over the synodic cycle.34 A DNA structure linked to the accumulation of carbohydrates is more developed at the new moon, whereas a structure appropriate for growth and flowering is more marked at the full moon. Mitotic activity is at its maximum at the first and last quarters, alternating with minima at the full and new moon. However, the researchers find certain types of cells reproducing out of step with this rhythm.
• Various algae follow lunar periodicities in their reproduction.35 This is expressed, for example, through the level of alginic acid in the brown alga Fucus virsoides.36 Similarly, the cytokinins, a group of plant growth hormones, vary with maxima at different lunar phases in the algae Ecklonia maxima and Sargassum heterophyllum.37
• In plants tested daily, such as the common bean, Philodendron (Philodendron sagittifolium) or geranium (Geranium spp), Rounds (1982) identifies a semi-lunar periodicity in the synthesis of acetylcholine or noradrenaline, which can be measured by the effect on the cardiac rhythm of the American cockroach Periplaneta americana.38 These observations illustrate the finding that the moment of harvest of medicinal plants has an impact on the pharmacological effect. This suggests once again the validity of certain traditional practices concerning lunar phases that were current in monasteries producing this type of plant.
• A reinterpretation of old systematic records of the movement of leaves of the sword-bean (Canavalia ensiformis) in a stable environment—carrying on the work of the founder of chronobiology, E. Bünning—allows P. Barlow, by calculating the corresponding gravimetric tides, to reveal a clearly marked tidal rhythm.39
• The variations in gaseous exchange observed by Graviou on tomato seeds in apparent dormancy, kept in constant light and temperature conditions, had shown maximal absorption of oxygen at full moon and new moon.40 This result agrees with the discovery by Brown and his collaborators that the potato and the carrot follow a synodic lunar rhythm in their respiration, with the carrot's oxygen consumption at a maximum for the 3rd quarter and the new moon, and at a minimum for the full moon.41 For the potato, a daily lunar rhythm is superposed onto this monthly rhythm: laboratory records maintained for 10 years in constant dark conditions, show a maximum at moonrise and culmination, but only from September to February and not the rest of the year. Graviou subsequently found a similar rhythm in the respiration of sunflower seeds (Helianthus annuus).
• Laboratory trials with cress over 3 years allowed Maw to establish that growth was accelerated in winter at the new moon and slowed at the full moon for plants subjected to a negatively ionized atmosphere, while these effects occurred in summer at the first and last quarters.42 On the other hand, no such periodicity is observed when the ionization is positive. The polluted air of towns being positively ionized compared to the pure air of a forest, Plaisance suggested multiple prospects of benefits of the negatively charged forest atmosphere for body and spirit.43 Might this be due to a more marked effect of lunar rhythms?
• It is possible that rhythmic differences in the speed and rate of germination are linked to cyclic variations in the absorption of water by the seeds, as Brown and Chow (1983) showed using an impressive experimental set-up (7,931 series of 20 beans).44 The seeds immersed in water day after day for 4 hours absorb water according to a circaseptan synodic lunar cycle (period of 7.4 days). It is remarkable that the variations in absorption observed over half a lunar week are so considerable, up to 20%. This suggests that it could be a matter here of variations in the properties of water, fluctuations which had already been discovered experimentally. Through series of standardized chemical reactions (tests of precipitation of bismuth chloride), Piccardi and Cini (1960) had demonstrated this phenomenon in experiments extending over more than 30 years.45 This phenomenon had already been verified by Burkard (1955), who showed the role of variations in atmospheric pressure, which are known to follow the rhythm of the moon cycles—hence the expression “atmospheric tides.”46 It was then analyzed statistically by De Meyer and Capel-Boute on the basis of long complementary series.47
• The variations in water absorption by seeds according to synodic rhythms were confirmed by Innamorati and Signorini (1980), then by Spruyt and others (1987), as an addition to the seasonal variation.48,49 Strangely enough, these last authors, on the basis of 25 lunations (2 years) with tests of daily immersion in constant laboratory conditions, also obtain a circaseptan lunar rhythm, but out of phase with that of Brown & Chow (1973; see Figure 4).50
• Abrami (1972) shows another lunar periodic phenomenon influencing the growth of plants.51 This research carried out outdoors concerns the role of the “temperature” factor on the elongation of the shoot of 7 wild herbaceous annual species: snowdrop (Galanthus nivalis), hollowroot (Corydalis cava), wood anemone (Anemone nemorosa), tuberous comfrey (Symphytum tuberosum), bear garlic (Allium ursinum), ground elder (Aegopodium podagraria), and creeping bellflower (Campanula rapunculoides). Obviously, growth is slower in cold weather and faster in hot weather; however, these experiments show that the correlation between these 2 variables fluctuates periodically, and differently depending on the species, according to rhythms of 29.5, 14.7, 9.7, and 7.3 days. Thus, the rate of growth of the bellflower is more strongly influenced by temperature in the new moon phase than in the full moon phase in March, April, and May.
• Concerning the factor “light,” and contrary to Semmens' (1923) observations,52 Kolisko and Kolisko (1939, 1953) had noted in the germination trials carried out at different depths in the soil that the synodic lunar rhythms persisted. This would seem to imply that a factor complementary to the polarized light is at work.53
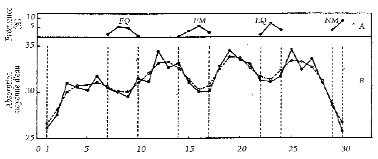 Figure 4: Variations in the absorption of water by bean seeds after 4 hours' soaking in constant laboratory conditions (25°C, darkness), relative to the lunar phases during 25 lunations (from June 1984 to July 1986). Values in % of initial dry weight. NM=New Moon / FM=Full Moon / FQ = first quarter / LQ = last quarter. Dotted line: moving averages over 3 days. Top of graph (A): position of the 4 phases in the average lunar cycle.2
Woody Plants
Woody plants also reveal in their physiology rhythmic phenomena linked with the moon:
• A study of secondary plant chemistry seeking to test a phytopractice native to Central America—according to which, the harvesting of leaves to make roofs occurs in relation to lunar phases—was carried out by Vogt and others (2002).54 Significant differences in carbon content (in different forms), calcium content, and the hemicellulose fraction could, according to these authors, explain the better durability in the full moon phase. However, marked differences are observed between the leaves of the 3 species analyzed: palma de sierra (Prestoea montana), lobster claw (Heliconia caribaea) and palo de cachinibo, yellow palicourea (Palicourea riparia). Note that the intermediary periods that proved to be interesting in other studies, such as the days between the first quarter and the full moon, were not taken into consideration here.
• When a pluridisciplinary team took up the results of work already published by Cantiani et al. (1994)55 on the variations in diameter of trees kept in constant conditions, they demonstrated the existence of a synodic lunar rhythm at a daily level, corresponding to gravimetric tides. Behind the photo-thermoperiodic cycle of 24 hours known for most physiological processes and corresponding to the influence of the sun, Zürcher et al. (1998) found that the diameter of tree-trunks also changes according to a lunar cycle with a period of 24.8 hours.56 This raises the question about the processes leading to these reversible fluctuations in diameter, and suggests that it could be an alternation in the relative water content of the cell wall compared to the cytoplasm.
• This last lunar phenomenon has recently been elegantly confirmed and had new light thrown on it, following on from Burr's work stretching from 1944 to 1972.57 Holzknecht presents an extremely sensitive device to measure bio-electric potentials of standing trees, applied to spruce and Swiss stone pine (P. cembra), which enables the detection of rhythms in phase with gravimetric daily tides and with the synodic lunar monthly cycle, during the trees' rest period in the winter.58 On the other hand, the ordinary 24-hour period predominates while the trees are growing.59
• In contrast to the potato,60 in spruce, the mitotic activity of polyembryonic tissue kept in constant laboratory conditions goes through maxima at full and new moon and minima at the first and last quarter.61
• Just recently, a new overhaul of Cantiani's 1994 data already analyzed by Zürcher et al. in 1998, and an in-depth analysis of other data of the Florence researchers about trees living in open conditions, enabled a spectacular confirmation of the role of gravimetric tides in tree physiology.62 These same authors also put forward the hypothesis of a possible role of geomagnetic field activity.
The Cosmic Dance of the Buds
Lunar rhythms in the shape of organs, analogous to those of the trunk diameters described above, had already been discovered by Edwards (1982) thanks to a meticulous series of photographic observations of tree buds.63 With the help of a shape factor developed in projective geometry, each bud—whether it be spherical, elliptical, or ovoid—can be characterized by a unique parameter Lambda. This shape, therefore lambda, not only changes radically at budbreak, but varies subtly around the characteristic value all through the bud's existence, from formation to budbreak. The phenomenon consists of a rhythmical elongation and relaxation of the buds all through the winter, as though there were breathing or a subtle heartbeat, already hinting at tiny movements of opening and closing. Edwards also indicates that this fluctuation in shape is synchronized for some species with the position of the moon relative to the sun (synodic rhythm), but for others with the alignment of the moon with certain planets, for example, Saturn for beech (Fagus sylvatica) and Mars for oak.
For mistletoe (Viscum album), a hemi-parasite of certain deciduoustrees and conifers, the analysis with the shape factor according to Edwards was applied to translucent berries.64,65 The measures of berries harvested over 6 years, between 1991 and 2001, revealed reversible fluctuations in an amplitude going from Lambda = 0.8 to Lambda = 1.2. These variations show here a significant correlation with the position of the moon relative to the fixed Zodiac constellations (sidereal rhythm). These results agree with the limits between the constellations mentioned since ancient times, and with their groupings mentioned in relation to M. Thun's work.
Felling Dates and Wood Properties
To return to forestry traditions, and to the codes about choosing felling dates according to the season and lunar cycles, we shall now tackle an area linked to a particular methodology, since it is now a matter of whole trees.
Samples to test and analyze must come both from the partly living sapwood and from the heartwood or “perfect wood,” much dryer and devoid of active cells: this part constitutes most of the volume of the bole for adult trees. The phenomena and processes involved are therefore mostly physical, and partly biological for the sapwood samples.
Apart from the 1930's research on bamboo (Dendrocalamus strictus) by Beeson and Bhatia (1936), indicating a water content that increases from full to new moon and decreases from new to full moon,66 it is only recently that the question of how felling trees according to the moon may influence the properties of the wood has been scientifically studied.67,68,69,70,71
Three separate studies were based on 6 specific dates, each supposed to represent 3 “favorable” felling dates alternating with 3 “unfavorable” dates. The number of spruce trees was 120, 60, and 30, respectively. The authors' opinions differ concerning the statistical treatment and the interpretation of the results, although the variations in density after drying coincided.71 In order to deal with the question more fundamentally and with a large data base, a new trial was conducted simultaneously in 4 sites in Switzerland, with 48 successive fellings (each Monday and Thursday)—with no link to an experimental hypothesis—of 3 trees per site over 5.5 months, representing a total of 624 trees felled over the winter from 2003 to 2004. The species used were spruce and sweet chestnut. Each tree provided a series of samples of the sapwood and heartwood, from different parts of the bole. The behavior of this material during drying in standard conditions was monitored. Among the different rhythms observed and statistically confirmed for 3 principle criteria was water loss, which for spruce varied systematically between the fellings just before the full moon and those just after.72 This type of variation is probably due not to differences in initial water content, but to the fact that the forces binding water to the cell walls of the ligneous tissue could be subject to fluctuations. The ratio between water easily extractible from the wood, called “free,” and that extracted below the saturation point of the fibers, or “bound water,” fluctuates according to the lunar cycles, and probably also according to the seasons. It should be noted that the characteristics of the rhythms are species-specific.
The statistical analysis indicates not only different synodic rhythms, but unexpectedly also a marked sidereal rhythm. The research thus supports the hypothesis that the “lunar practices” of foresters are based on a kernel of objective observations.
Implications and Prospects
This overview of lunar cycles found in the plant world reveals a real phenomenon that is additional to the main exogenous rhythms of solar origin, the action of which is well known, on a daily as much as on a seasonal level, and even that linked to the activity cycle of sunspots with a period of around 11 years. The moon modulates this main exogenous rhythm on an hourly scale, the gravimetric tides taking place with 2 ebbs and flows a day, as well as on the level of the lunar week and month, following the synodic, tropical, sidereal, or anomalistic (with perigee and apogee) cycles. It seems that lunar rhythms become apparent when the sun’s influence is withdrawn, be it naturally or with the help of an experimental device.
There is now the issue of the nature of the forces involved. Regarding the synodic and anomalistic rhythms, the gravitational force that causes the tides is too weak to explain even part of the lunar phenomena observed on plants: it is no more than 80 billionths of the force exerted by gravity on an object on the Earth’s surface.73
For the biggest tree measured in Europe74—a silver fir (A. alba) of the Black Forest with a height of 68 meters, a diameter of 380 cm, a trunk volume of 140 m3, and a weight of 100 tons—the tidal force of the moon represents a daily pull and release of only 8g—the weight of 2 sugar cubes!
The weak yet distinct variations of the geomagnetic field, with a period of a half lunar day (12h 25min) and caused by the gravimetric tides, present an analogous case. Baillaud (2004) comments rightly: “In lunar matters, the sympathiser […] will wish to be shown the medium of the link between the Moon and the living being and that the sequence of phenomena be dissected for him, or at least a hypothesis be put forward.”73
It becomes clearer, as has been mentioned several times, that this medium is none other than the essential element for any organic process, i.e., water. This idea was supported by the conclusions of Piccardi (1962),75 confirmed by Eichmeier & Büger (1969),76 and those of Tromp (1972),77 as the famous Austrian philosopher and science theorist Paul Feyerabend (1978) had underlined in Science in a Free Society, bringing to bear his abilities in mathematics, physics, and astronomy.78
Already in the 1920's, experiments had been carried out on the variations of the surface tension of water using extremely thin glass capillary tubes.79 They were able to show that lunar rhythms (daily tidal rhythms among others) started appearing at a certain thinness of the capillaries. Meanwhile, it was established that being in a capillary system, whether glass or organic such as plant cells (with their cavities, but also their finely porous walls), significantly modifies the properties of water, such as the ability to stay liquid at temperatures reaching -15°C.80 It would be interesting to analyze once again the effect of the time factor on these essential properties of water using modern-day technology.
A recent double publication in theoretical physics by Dorda (2004), the co-author with von Klitzing of the discovery of the Quantum Hall Effect that led to the 1985 Nobel Prize in Physics, puts forward a new astrogeophysical model of the role of gravitation in living processes.81 This model integrates the static and dynamic aspects of gravitation relative to the orbital movement of celestial bodies, leads to a “quantization” of gravitation and time, and demonstrates a reversible effect linked partly to the sun and partly to the moon on the supra-molecular structure of water. Thus, the model leads to the determination of reversible aggregation or coherence states (clusters) of water, in a quantitative relation of considerable amplitude of variation, from 1 to 2,200, depending on whether the interaction is sun-Earth or moon-Earth, this last interaction modulating according to the lunar day but also according to the phase (waxing or waning). Dorda considers that this rhythmic fluctuation of water in a system of 3 celestial bodies constitutes the clock long sought in organic structures. This model was able to be validated independently using experimental measurements already published and interpreted along the same lines by the teams of Cantiani and Zürcher in 1994 and 1998.82,83
Finally, Vallée (2004) developed a new and reproducible experimental method that proves that weak and low-frequency electromagnetic fields have a durable effect on water.84 This researcher stresses the importance of the interfaces between water and its solid or gaseous inclusions—an essential aspect, since interfacial water plays a fundamental role in the organic world.
Rossignol et al. (1998) emphasize the role of electromagnetic phenomena linked to lunar cycles (polarization of light, atmospheric pressure) and consider there may be a link with the induction of bio-electric potentials at the cell level.85
Nevertheless, all these discoveries and interpretations on a physical level do not explain why such differences are observed between certain living plant species, annual or woody.
In parallel to these hypotheses emanating from fundamental research, taking lunar chronobiology into consideration in fieldwork with plants opens up prospects that, though unexpected, were already suggested by certain traditional practices:
• Plant production and selection respecting the fundamentally rhythmic nature of each species, which, for example, enabled Martin Schmidt to develop between 1944 and 1964 a new variety of rye still cultivated today.86 The method takes into account not only rhythmic aspects, but also the position of the grains on the ear's axis, similarly to the “Phytopractices” in tropical regions.87
• Reforestations of superior quality, with plants robust against diseases, deriving from nurseries obtaining high levels of germination, with “carbon forestry and agroforestry” as a prospect.88
• An ecological and bio-compatible wood technology, thanks to chronobiologically correct felling, if long-term research confirms and completes the already obtained results.
• A plant pharmacology making the most of the cycles of efficacy of the active principles.
Considering the “time” factor as an essential environmental component enables biotechnologies in the true sense of the term to be developed, bringing organisms—in this case, plants and their specific constituents—to the full expression of their potential.
Professor Ernst Zürcher, PhD, currently lectures in wood science at the Swiss Federal Institute of Technology in Lausanne, and at the Bern University of Applied Sciences in Biel, Switzerland. He trained as a forestry engineer in Zürich and obtained his doctorate there on tree morphology and wood anatomy. Subsequent research activities included tree vitality, spiral grain, and chronobiology. He spent 4 years in Rwanda with the Swiss Development Cooperation, and has since led research projects on temperate and tropical wood biology, physics and technology, natural wood protection, and on the role of felling date on wood properties. He can be contacted at: ernst.zuercher@bfh.ch.
Illustration References
1. ROHMEDER, E. (1938): Der Einfluss der Mondphasen auf die Keimung und erste Jugendentwicklung der Fichte. Forstwissenschaftliches Zentralblatt 60.Jhrg, Heft 19 (1938): 593-603; 634-646.
2. SPRUYT, E., VERBELEN, J.-P., DE GREEF, J. A. (1987): Expression of Circaseptan and Circannual Rhythmicity in the Imbibition of Dry Stored Bean Seeds. Plant Physiol. 84: 707-710.
References
1. GERBER, E., Schmalz, K. L. (1948): Findlinge. Berner Heimatbücher Nr. 34. Haupt, Bern.
2. MORAN UBIDIA, J. (2003): Traditional Bamboo preservation methods in Latin America. Faculty of Architecture and Urbanism, University of Guayaquil, Ecuador.
3. SCHMIDT, C. (1940) Ed.: Manichäische Handschriften. (95 Erste Kapitel). Verlag Kohlhammer, Stuttgart.
4. Serres, O. de (1600): Le Théâtre d'agriculture et mesnage des champs. Jamet-Métayer, Paris (1400 p).
5. Endres, K.-P., Schad, W. (1997): Biology of the Moon. Moon periodicities and Life Rhythms. (in German) S.Hirzel verlag, Stuttgart / Leipzig.
6. ENGELMANN, W. (2004): Rhythms of Life. An introduction using selected topics and examples. University of Tübingen.
7. PEARSE, J.S. (1990): Lunar reproductive rhythms in marine invertebrates: maximizing fertilization? In: Advances in Invertebrate Reproduction 5. Hoshi, M., Yamashita, O. (eds): 311-316. New York, Elsevier Science.
8. PALMER, J.D. (1995): The Biological Rhythms and Clocks of Intertidal Animal. Oxford University Press, New York.
9. BRACHER, R. (1938): The light relations of Euglena limosa Gard. Part 1: The influence of intensity and quality of light on phototaxy. Journal of the Linnean Society, Botany 51: 23-42.
10. SEMMENS, E. (1923): Effect of Moonlight on the germination of Seeds. Nature Vol. 111: 49-50.
11. EMMENS, E. (1947): Chemical Effects of Moonlight. Nature No. 4044, May 3, 1947: p.613.
12. Kolisko, L. (1927): The moon and the plant growth. (in German) In Wachsmuth, G. (ed), “Gäa-Sophia.” Jahrbuch der naturwissenschaftlichen Sektion am Goetheanum. Dornach, Switzerland. Bd II: 358-379.
Kolisko, L. (1929): The moon and the plant growth. (in German) In Wachsmuth, G. (ed), “Gäa-Sophia.” Landwirtschaft. Dornach, Switzerland. Bd IV: 84-94.
Kolisko, L. (1934): The moon and the plant growth. (in German) Mitteilungen des Biologischen Institutes am Goetheanum. Dornach, Switzerland. Nr.1: 19-21 and Nr.2: 17-24.
Kolisko, L. (1935): The moon and the plant growth. (in German) Mitteilungen des Biologischen Institutes am Goetheanum. Dornach, Switzerland. Nr.3: 17-19 and Nr.4: 3-14.
Kolisko, E. and L. Kolisko. (1939 / 1953): Agriculture of Tomorrow, Stroud, Gloucester, England: Kolisko Archive, original publication, 1939, Bournemouth 1947 Trad. allem.: Landwirtschaft der Zukunft, Meier & Cie, Schaffhausen Switzerland, 1953.
13. STEINER, R. (1924) : Agriculture, Fondements spirituels de la méthode Bio-dynamique (9 conferences). Traduction 1974, Editions Anthroposophiques Romandes, Genève.
14. VOEGELE, L. (1930): Dynamische Wirkungen und ihre praktische Auswertung. Demeter 5 (12): 241-245.
15. POPP, M. (1933): Hat der Mond einen Einfluss auf das Pflanzenwachstum? Z. Pflanzenernährung, Düngung, Bodenkunde Bd.11 (4): 145-150.
16. POPP, M. (1936): Hat der Mond einen Einfluss auf das Pflanzenwachstum? 2. Mitt. Z. Pflanzenernährung, Düngung, Bodenkunde 41: 348-357.
17. POPP, M. (1937): Hat der Mond einen Einfluss auf das Pflanzenwachstum? 3. Mitt. Z. Pflanzenernährung, Düngung, Bodenkunde 48 (3/4): 133-138.
18. OPITZ, K. (1936): Über den Einfluss des Mondes auf das Pflanzenwachstum. Z. Pflanzenernährung, Düngung, Bodenkunde 41: 357-359.
19. BREDA, E. (1960): Untersuchungen von Saatgut- und Nahrungsqualität in Abhängigkeit vom Jahresrhythmus. Forschungsbericht, Inst. Biol.-dynam. Forsch., Darmstadt (noto published).
20. ABELE, U. (1973): Vergleichende Untersuchungen zum konventionellen und biologisch-dynamischen Pflanzenbau unter besonderer Berücksichtigung von Saatzeit und Entitäten. Dissertation. Justus Liebig-Universität Giessen. Inst. für Pflanzenbau und Pflanzenzüchtung.
21. ABELE, U. (1975): Saatzeitversuch mit Radies. Lebendige Erde 6: 223-225.
22. Spiess, H. (1994): Chronobiologische Untersuchungen mit besonderer Berucksichtigung lunarer Rhythmen im biologische-dynamischen Pflanzenbau, Darmstadt; 2 vols.
23. POPP, M. (1933): Hat der Mond einen Einfluss auf das Pflanzenwachstum? Z. Pflanzenernährung, Düngung, Bodenkunde Bd.11 (4): 145-150.
24. KOLLERSTROM, N., STAUDENMAIER, G. (2001): Evidence for Lunar-Sidereal Rhythms in Crop Yield: A Review. Biological Agriculture and Horticulture, Vol. 19: 247-259.
25. FRITZ, J. (1994): Untersuchungen zum Einfluß des synodischen Mondrhythmus auf das Pflanzenwachstum von Rettich (Raphanus sativus) und Efeuaralie (Fatshedera lizei). Witzenhausen (Dipl.-Arbeit). Universität Gesamthochschule Kassel.
26. MILTON, W.J. (1974): Exogenous variations in plant (Zea mais) germination and growth in darkness and ‘constant’ temperature, modified by uniform daily rotations. Dissertation, Northwestern University, Evanston, Illinois.
27. MILLET, B. (1970): Analyse des rythmes de croissance de la Feve. {Vicia faba L.). (Thèse Ph.D) Ann. Sci. Univ. Besancon, 3eme ser., Bot. 8:. 1-132.
28. GRAVIOU, E. (1977): Variabilité non aléatoire d’un matériel végétal dans ses échanges gazeux et dans sa croissance. Bulletin Mensuel de la Société Linnéenne de Lyon 46(4) : 108-112.
29. Zürcher, E. (1992): Rhythmicities in the Germination and Initial Growth of a Tropical Forest Tree Species. (in French with a German summary) Schweizerische Zeitschrift für Forstwesen, Journal Forestier Suisse, 143(1992):951-966.
30. BAGNOUD, N. (1995): Rythmicités dans la germination et la croissance initiale de 4 essences ligneuses de la Zone Soudano-Sahélienne. Essai lunaison. Groupe de foresterie pour le développement, IER Sikasso, Mali / Intercoopération, Bern (13 p).
31. BEESON, C. F. C. (1946): The moon and plant growth Nature. 158: 572-573.
32. ROHMEDER, E. (1938): Der Einfluss der Mondphasen auf die Keimung und erste Jugendentwicklung der Fichte. Forstwissenschaftliches Zentralblatt 60.Jhrg, Heft 19 (1938): 593-603; 634-646.
33. Zürcher, E. (2008): Lunar-synodic variation in the germination of European spruce (Picea abies) seeds: a previous trial re-evaluated (in preparation).
34. Rossignol, M., Benzine-Tizroutine, S., Rossignol, L. (1990): Lunar cycle and nuclear DNA variations in potato callus or root meristem. In Tomassen, G.J.M., de Graaff, W., Knoop, A. A. and Hengeveld, R. (eds), Geo-cosmic relations; the earth and its macro-environment. Proceedings of the First International Congress on Geo-cosmic Relations, April 19-22, 1989, Amsterdam. Pudoc, Wageningen, The Netherlands.
Rossignol, M., Rossignol, L., Oldeman, R., Benzine-Tizroutine, S. (1998): The Struggle of Life - Or the Natural History of Stress and Adaptation. Grafish Service Centrum Van Gils b.v., Wageningen, NL.
35. Endres, K.-P., Schad, W. (1997): Biology of the Moon. Moon periodicities and Life Rhythms. (in German) S.Hirzel verlag, Stuttgart / Leipzig.
36. LAUSI,D., DE CRISTINI, P. (1967): Osservazioni sulle variazioni periodiche del contenuto in acido alginico in Fucus virsoides. Nova Thalassia (Trieste) 3(4): 1-16.
37. HOFMAN, P.J., FEATONBY-SMITH, C. B., VAN STADEN, J. (1986): The development of ELISA and IRA for cytokinin estimation and their application to a study of lunar periodicity in Ecklonia maxima (Osbeck) Papenf.. J. Pl. Physiol. 122: 455-466.
38. ROUNDS, H.D. (1982): A semilunar periodicity of neurotransmitter-like substances from plants. Physiol. Plantarum 54 (4): 495-499.
39. KLEIN, G. (2007): Farewell to the Internal Clock. A contribution in the field of chronobiology. Springer, New York (116 p) - Preface.
40. GRAVIOU, E. (1978): Analogies between rhythms in plant material, in atmospheric pressure, and solar lunar periodicities. International Journal of Biometeorology. Vol.22 (2): 103-111.
41. BROWN et al. (1954, 1955, 1962, 1965): cf. Spiess 1994 – ref. 22.
42. MAW, M.G. (1967): Periodicities in the influences of air ions on the growth of garden cress, Lepidum sativum L. Canadian Journal of Plant Science 47: 499-507
43. PLAISANCE (1985): Forêt et Santé. Guide pratique de sylvothérapie. Editions Dangles, Saint-Jean-de-Braye (F).
44. BROWN, F. A., Chow, C. S. (1973): Lunar-correlated Variations in Water Uptake by Bean Seeds. Biol. Bull. 145: 265-278.
45. PICCARDI, G., CINI, R. (1960): Polymerization and the low-frequency electromagnetic field. J. of Pol. Sci., 48, 393 (1960).
46. BURKARD, O. (1955): Einige statistische Versuche zur Piccardischen Fällungsreaktion. J. Theoretical and Applied Climatology, Vol.6 (4): 506-510.
47. DE MEYER, F., CAPEL-BOUTE, C. (1987): Statistical analysis of Piccardi chemical tests. International Journal of Biometeorology, Vol. 31 (4): 301-322.
48. INNAMORATI, M., SIGNORINI, P. (1980): Ritmi nei vegetali: rilevamento ed analisi. G. Bot. Italiano 114 (3-4): 124.
49. SPRUYT, E., VERBELEN, J.-P., DE GREEF, J. A. (1987): Expression of Circaseptan and Circannual Rhythmicity in the Imbibition of Dry Stored Bean Seeds. Plant Physiol. 84: 707-710.
50. BROWN, F. A., Chow, C. S. (1973): Lunar-correlated Variations in Water Uptake by Bean Seeds. Biol. Bull. 145: 265-278.
51. ABRAMI, G. (1972): Correlations between lunar phases and rhythmicities in plant growth under field conditions. Can. J. Bot. 50: 2157-2166.
52. SEMMENS, E. (1923): Effect of Moonlight on the germination of Seeds. Nature Vol. 111: 49-50.
53. Kolisko, E. and L. Kolisko. (1939 / 1953): Agriculture of Tomorrow, Stroud, Gloucester, England: Kolisko Archive, original publication, 1939, Bournemouth 1947 Trad. allem.: Landwirtschaft der Zukunft, Meier & Cie, Schaffhausen Switzerland, 1953.
54. Vogt, K. A., Beard, K. H., Hammann, S., O'Hara Palmiotto J., Vogt, D. J., Scatena, F. N., Hecht, B. P. (2002): Indigenous Knowledge Informing Management of Tropical Forests: The Link between Rhythms in Plant Secondary Chemistry and Lunar Cycles. Ambio Vol. 31 No. 6, Sept 2002: 485- 490.
55. Cantiani, M. (†), Cantiani, M.-G., Sorbetti Guerri, F. 1994: Rythmes d'accroissement en diamètre des arbres forestiers. Revue Forestière Française 46: 349-358.
56. Zürcher, E., Cantiani, M.-G., Sorbetti-Guerri, F., Michel, D. (1998): Tree stem diameters fluctuate with tide. Nature 392: 665-666.
57. Burr, H.S. (1944): Moon-Madness. Yale Journal of biology and medicine 16: 249-256.
Burr, H.S. 1945. Diurnal Potentials in the Maple Tree. Yale J. Biol. and Med. 17(6):727-734.
Burr, H.S. 1947. Tree Potentials. Yale J. Biol. and Med. 19(3):311-318.
Burr, H.S. 1972: Blueprint for Immortality: The Electric Patterns of Life. C. W. Daniel Company Ltd, Saffron Waldon, Essex, England.
58. Holzknecht, K. (2002): Elektrische Potentiale im Splintholz von Fichte und Zirbe im Zusammen¬hang mit Klima und Mondphasen / Electrical potential in the sapwood of Norway spruce (Picea abies L.) and stone pine (Pinus cembra L.) and their relationship with climate and lunar phase. Universitaet Innsbruck , Naturwissenschaftliche Fakultaet, Institut fuer Botanik. PhD-Thesis G0443 Physiol.
59. Holzknecht, K., Zürcher, E. (2006): Tree stems and tides - A new approach and elements of reflexion. Schweiz. Z. Forstwes. 157 (2006) 6: 185-190; figure 6.
60. See reference 34
61. VLASINOVA, H., MIKULECKY, M., HAVEL, L. (2003/4): The mitotic activity of Norway spruce polyembryonic culture oscillates during the synodic lunar cycle. Biologia Plantarum 47 (3): 475-476.
62. BARLOW, P. W., MIKULECKY, M., STRESTIK, J. (2000): Tee-stem diameter fluctuates with the lunar tides and perhaps with geomagnetic activity. Protoplasma 2010, Apr. 15 (published online).
63. EDWARDS, L. (1982): The field of form. Floris Books, Edinburgh.
EDWARDS, L. (1993): The Vortex of Life - Nature’s patterns in space and time. Floris Books, Edinburgh.
64. FLÜCKIGER, H., BAUMGARTNER, S. (2002): Formveränderungen reifender Mistelbeeren. Elemente der Naturwissenschaften, 77(2/2002): 2-15.
65. BAUMGARTNER, S., FLÜCKIGER, H. (2004): Mistelbeeren - Spiegel von Mond- und Sternbild-Konstellationen. Mistilteinn, vol. 5: 4-19.
66. BEESON, C.F.C. & BHATIA (1936) quoted in BEESON (1946) cf. Ref. 31.
67. Triebel, J. (1998): Mondphasenabhängiger Holzeinschlag - Literaturbetrachtung und Untersuchung ausgewählter Eigenschaften des Holzes von Fichten (Picea abies Karst.). Diplomarbeit, Institut für Forstbenutzung und Forsttechnik, TU Dresden (108 p).
68. Seeling, U., Herz, A. (1998): Einfluss des Fällzeitpunktes auf das Schwindungsverhalten und die Feuchte des Holzes von Fichte (Picea abies) - Literaturübersicht und Pilotstudie. Albert-Ludwigs-Universität Freiburg i.Br., Institut Forstbenutzung und Forstliche Arbeitswissenschaft. Arbeitspapier 2-98 (66p).
69. Seeling, U. (2000): Ausgewählte Eigenschaften des Holzes der Fichte (Picea abies (L.) Karst.) in Abhängigkeit vom Zeitpunkt der Fällung. Schweizerische Zeitschrift für Forstwesen 151 (2000) 11: 451-458.
70. BARISKA, M., RÖSCH, P. (2000): Fällzeit und Schwindverhalten von Fichtenholz. Schweizerische Zeitschrift für Forstwesen 151 (2000) 11: 439-443.
71. Zürcher, E., Mandallaz, D. (2001): Lunar synodic Rhythm and Wood Properties: Traditions and Reality. In: L'arbre 2000 The Tree. 4th International Symposium on the Tree, 20-26 August, 2000. Institut de recherche en biologie végétale / Montréal Botanic Garden, Isabelle Quentin Editeur, Montréal: 244-250.
72. Zürcher, E., Schlaepfer, R., Conedera, M:, Giudici, F. (2010): Looking for differences in wood properties as a function of the felling date: lunar phase-correlated variations in the drying behavior of Norway Spruce (Picea abies Karst.) and Sweet Chestnut (Castanea sativa Mill.). TREES (2010) 24 : 31-41.
73. BAILLAUD, L. (2004): Chronobiologie lunaire controversée: de la nécessité de bonnes méthodologies. Bulletin du Groupe d’Etude des Rythmes Biologiques (35) 3 : 3-16.
74. KLEIN, L. (1908): In Mayer, H. Waldbau auf soziologisch-ökologischer Grundlage. G. Fischer, Stuttgart - Jena - New York, 1977. (S.51)
75. PICCARDI, G. (1962): The Clinical Basis of Medical Climatology. Charles. C. Thomas Publ., Springfield Ill.
76. EICHMEIER, J., BÜGER, P. (1969): Über den Einfluss elektromagnetischer Strahlung auf die Wismutchlorid—Fällungsreaktion nach Piccardi. International Journal of Biometeorology. Vol. 13 (3-4): 239-256.
77. TROMP, S.W. (1972): Possible effects of Extra-terrestrial stimuli on colloidal systems and living organisms. From: Biometeorology Volume 5 part II, Proceedings of the 6th International Biometeorological congress held at Noordwijk, The Netherlands 3-9 Sept 1972 S.W.Tromp, W.H.Weihe & Janneke J.Bouma. Supplement to Vol 16 1972 of The International Journal of Biometeorology.
78. FEYERABEND, P. (1978): Science in a Free Society. Doubleday, New York.
79. MAAG, G.W. (1928): Planeteneinflüsse. West-Ost-Verlag, Konstanz (215 p).
80. SPARKS, J. P., CAMPBELL, G.S., BLACK, R. A. (2000): Liquid water content of wood tissue at temperatures below 0°C. Can. J. For. Res. 30: 624-630.
81. DORDA, G. (2004): Sun, Earth, Moon - the Influence of Gravity on the Development of Organic Structures. Part II: The Influence of the Moon. Sudetendeutsche Akademie der Wissenschaften und Künste, München. Bd 25: 29-44.
82. Cantiani, M. (†), Cantiani, M.-G., Sorbetti Guerri, F. 1994: Rythmes d'accroissement en diamètre des arbres forestiers. Revue Forestière Française 46: 349-358.
83. Zürcher, E., Cantiani, M.-G., Sorbetti-Guerri, F., Michel, D. (1998): Tree stem diameters fluctuate with tide. Nature 392: 665-666.
84. VALLEE, PH. (2004): Etude de l’effet de champs électromagnétiques basse fréquence sur les propriétés physico-chimiques de l’eau. Thèse de Doctorat, Université Pierre et Marie Curie (Paris VI).
85. Rossignol, M., Rossignol, L., Oldeman, R., Benzine-Tizroutine, S. (1998): The Struggle of Life - Or the Natural History of Stress and Adaptation. Grafish Service Centrum Van Gils b.v., Wageningen, NL.
86. FRANCKE, H. (2001): Das Ährenbeet von Martin Schmidt. Lebendige Erde 3/2001: 40 – 43. [voir aussi G. W. Schmidt „In-situ-Erhaltung pflanzengenetischer Ressourcen im ökologischen Landbau“ (Internet)]
87. AUMEERUDDY, Y., PINGLO, F. (1989): Phytopractices in tropical regions. A preliminary survey of traditional crop improvement techniques. Unesco/Man and the Biosphere Programme MAB, Institut de Botanique/Laboratoire de Botanique Tropicale, Montpellier. Published by the United Nations Educational, Scientific and Cultural Organization, 7, Place de Fontenoy, 75700 Paris (71 p).
88. VERCHOT, L.V., MACKENSEN, J., Kandji, S., van Noordwijk, M., Tomich, T. P., Ong, C. K., Albrecht, A., Bantilan, C., Anupama, K., Palm, C. A. (2005): Opportunities for linking adaptation and mitigation in agroforestry systems. In: Robledo C.; Kanninen M.; Pedroni L. (Ed.) 2005. Tropical forests and adaptation to climate change. In search of synergies. Center for International Forestry Research, CIFOR, Jakarta.
|