Issue: 56 Page: 40-52
Rhodiola rosea: A Phytomedicinal Overview
by Richard P. Brown, Patricia L. Gerbarg, Zakir Ramazanov
HerbalGram. 2002; 56:40-52 American Botanical Council
Rhodiola rosea: A Phytomedicinal Overview
Sidebar: "Clinical Case Studies R. rosea"
Rhodiola rosea: A Phytomedicinal Overview
by Richard P. Brown, M.D., Patricia L. Gerbarg, M.D., and Zakir Ramazanov, Ph.D., D.S.
Rhodiola rosea L.,
also known as "golden root" or "roseroot" belongs to the plant family
Crassulaceae.1 R.
rosea grows primarily in dry sandy ground
at high altitudes in the arctic areas of Europe and Asia.2 The plant
reaches a height of 12 to 30 inches (70cm) and produces yellow blossoms. It is
a perennial with a thick rhizome, fragrant when cut. The Greek physician,
Dioscorides, first recorded medicinal applications of rodia riza in 77 C.E. in De Materia Medica.3 Linnaeus renamed it Rhodiola
rosea, referring to the rose-like attar
(fragrance) of the fresh cut rootstock.4
For centuries, R. rosea has been used in the
traditional medicine of Russia, Scandinavia, and other countries. Between 1725
and 1960, various medicinal applications of R. rosea appeared in the scientific literature of Sweden,
Norway, France, Germany, the Soviet Union, and Iceland.2,4-12 Since
1960, more than 180 pharmacological, phytochemical, and clinical studies have
been published. Although R. rosea has been extensively studied as
an adaptogen with various health-promoting effects, its properties remain
largely unknown in the West. In part this may be due to the fact that the bulk
of research has been published in Slavic and Scandinavian languages. This
review provides an introduction to some of the traditional uses of R. rosea, its
phytochemistry, scientific studies exploring its diverse physiological effects,
and its current and future medical applications.
Rhodiola rosea in Traditional Medicine
Traditional folk medicine used R. rosea to increase physical endurance, work productivity,
longevity, resistance to high altitude sickness, and to treat fatigue, depression,
anemia, impotence, gastrointestinal ailments, infections, and nervous system
disorders. In mountain villages of Siberia, a bouquet of roots is still given
to couples prior to marriage to enhance fertility and assure the birth of
healthy children.2 In Middle Asia, R. rosea tea was the most effective treatment for cold and
flu during severe Asian winters. Mongolian doctors prescribed it for
tuberculosis and cancer.13 For centuries, only family members knew
where to harvest the wild "golden roots" and the methods of extraction.2
Siberians secretly transported the herb down ancient trails to the Caucasian
Mountains where it was traded for Georgian wines, fruits, garlic, and honey.
Chinese emperors sent expeditions to Siberia to bring back the "golden root"
for medicinal preparations.
Linnaeus wrote of R. rosea as an astringent and for the treatment of hernia, leucorrhoea (vaginal
discharge), hysteria, and headache.4,7 In 1755 R. rosea was included in the first Swedish Pharmacopoeia.
Vikings used the herb to enhance their physical strength and endurance.14
German researchers described the benefits of R. rosea for pain, headache, scurvy, hemorrhoids, as a
stimulant, and as an anti-inflammatory.15,16
In 1961, G.V. Krylov, a Russian botanist and taxonomist in
the Department of Botany at the Novosibirsk Branch of the Russian Academy of
Sciences, led an expedition to the cedar taiga in the Altai Mountains of
southern Siberia where he located and identified the "golden root" as Rhodiola
rosea.17 Extracts of the R.
rosea root were found to contain powerful
adaptogens. Research revealed that it protected animals and humans from mental
and physical stress, toxins, and cold.2,17 The quest for new
medicines to treat diseases such as cancer and radiation sickness, and to
enhance physical and mental performance, led to the discovery of a group of
phenylpropanoids that are specific to R. rosea. (See Phytochemistry section below.)
Geographical Distribution and Taxonomy of Rhodiola rosea
While Rhodiola as a
genus may have originated in the mountainous regions of Southwest China and the
Himalayas,18 botanists have established that various species of the
genus Rhodiola naturally display
a circumpolar distribution in mountainous regions in the higher latitudes and
elevations of the Northern Hemisphere. In Central and Northern Asia, the genus
is distributed from the Altai Mountains across Mongolia into many parts of
Siberia.19 According to Hegi, its distribution in Europe extends
from Iceland and the British Isles across Scandinavia as far south as the
Pyrenees, the Alps, the Carpathian Mountains and other mountainous Balkan
regions. Several varieties of Rhodiola species have also been identified across Alaska, Canada, and the
northern mountains of the continental United States.20 In fact, the
world database of botanical literature shows many citations identifying a broad
range of species of the genus Rhodiola, in some cases including R. rosea, in many diverse locations in northern latitudes (see Table 1).
The current taxonomical status of the genus Rhodiola has become quite complex. Before World War II, some
taxonomists separated different species of Rhodiola into an independent genus, belonging to the
subfamily Sedoidae.20
Then Rhodiola was reclassified as
a subgenus of the larger genus Sedum,
which contained about 10 species. In 1963 Hegi identified more than 50 species
of Rhodiola and re-established
them as a separate genus.20 Due to their morphological similarities,
they form a distinct Rhodiola
group.21 There are still differing opinions among specialists about
which new species should or should not be included in the genus
Rhodiola. The rationale and defining
criteria for the boundaries of the genus remain somewhat controversial. This is
not, in itself, necessarily counterproductive, since the acquisition of
botanical knowledge inevitably stimulates new understanding and insight,
creating the need for revised systems of classification. In the case of R.
rosea, however, this taxonomic ambiguity
may have unexpected and potentially negative consequences.
Popularizing a phytomedicinal plant like R. rosea can create confusion when the public is offered a
variety of "Rhodiola" products
using the general plant family name instead of the full botanical name of the
particular species. For example, products called "Rhodiola spp., Tibetan Rhodiola or Indian Rhodiola" may incorrectly imply equivalence with R.
rosea extract. Because of significant
species-dependent variation in phytochemistry and pharmacology, the use of "Rhodiola" as a general term is inaccurate and misleading. The
correct identification of all Rhodiola species according to precise and generally accepted botanical,
phytochemical, and genetic taxonomic criteria is not merely an abstract
intellectual exercise. It is critical for both scientific and
phytopharmacological accuracy, as well as for product labeling for the public.
Consumers may need professional guidance to avoid purchasing ineffective
brands, particularly those that do not provide full information, including the
complete botanical name of the plant species. Companies may change their
suppliers over time. Therefore, consumers should periodically check independent
sources of product evaluation, as well as requesting information about quality
control and content from manufacturers.
The pharmacological and medicinal properties of Rhodiola are species-dependent phenomena.22 Of all
the Rhodiola species, R.
rosea has been the predominant subject of
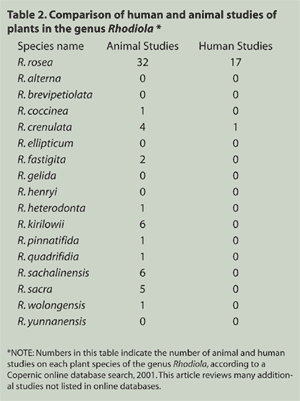
phytochemical, animal, and human studies.2,18,23,24 Table 2 compares
the research record of R. rosea
with all other species of the genus Rhodiola. Approximately 51 percent of all animal studies and
94 percent of all human studies conducted on plants in the genus Rhodiola are on the species R. rosea. Only R. rosea has passed extensive toxicological studies and has been certified safe
for both animals and humans.25

Phytochemistry of Rhodiola rosea
The investigation of the phytochemistry of R. rosea root has revealed the presence of six distinct
groups of chemical compounds:
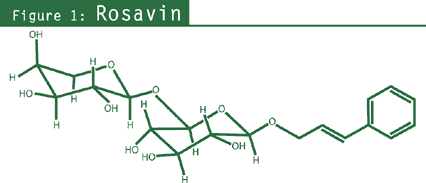
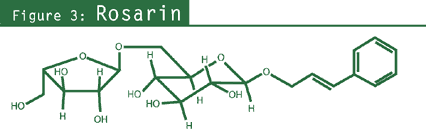
- Phenylpropanoids: rosavin, rosin, rosarin (specific to R. rosea;
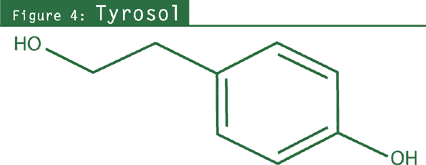
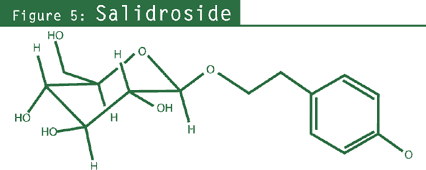
- Phenylethanol derivatives: salidroside (rhodioloside), tyrosol;
- Flavanoids: rodiolin, rodionin, rodiosin, acetylrodalgin, tricin;
- Monoterpernes: rosiridol, rosaridin;
- Triterpenes: daucosterol, beta-sitosterol;
- Phenolic acids: chlorogenic and hydroxycinnamic, gallic acids.
The standardization of R. rosea root extracts has gone through two distinct phases.
Initially, in the 1970s, the compound responsible for its unique pharmacological
properties was believed to be salidroside (rhodioloside).2,23,24,26,27
Therefore, the first generation of R. rosea tincture/extracts approved by the Russian
Pharmacopoeia Committee was standardized to a minimum of 0.8 percent
salidroside content.25
In the late 1980s, demand for R. rosea-based phytomedicines dramatically increased. The
wild-crafted raw material was over-harvested, resulting in a steady decline in
the quality and effectiveness of "Rhodiola" preparations.
Scientific investigation revealed that other species of genus Rhodiola
(which also contained salidroside) were
being substituted for R. rosea.
While some of these mixed batches were highly variable in quality, others had
no pharmacological effect. Logically, the suspicion arose that the salidroside
standard was inadequate. Based on comparative analysis, the obvious hypothesis
was that the original high potency product contained other active compounds
specific to R. rosea that had not
yet been identified.
Specific compounds set Rhodiola rosea apart from other Rhodiola species
After more than a decade of research, Kurkin and colleagues
presented evidence in 1986 that the chemical composition of R. rosea root is, in fact, different from the other species
of genus Rhodiola.23
Using newly developed methods of analysis, Dubichev and colleagues demonstrated
that R. rosea root contains three
cinnamyl alcohol-vicianosides — rosavin, rosin, and rosarin — that are specific
to this species.28,29 The term rosavins can be used to include rosavin, rosin, and rosarin
(see chemical figures).
It became evident that salidroside is present in all
chemically analyzed plants in the genus Rhodiola, and in a wide variety of species outside the genus.2,25-34
The term salidroside is derived
from Salix, the genus name for
the willows. Salidroside was first isolated in 1926 from Salix
triandra L. (Salicaceae).33
Since then it has been detected in Vaccinium vitis-idaea L. (Ericaceae) and in Rhododendron35,36 (plants not belonging to the genus
Rhodiola) in concentrations that can be
higher than levels found in Rhodiola species, including R. rosea.
Therefore, salidroside alone is not a useful marker compound for
differentiating true R. rosea
from other Rhodiola species; nor
should it be used as the only marker compound for the standardization of R.
rosea root extracts.
According to the revised 1989 Soviet Pharmacopeia,37
the extracts of R. rosea — primarily in
the form of water/alcohol tinctures or dried root extract — are now
standardized for both rosavins and salidroside.
Although rosavins are now the accepted marker for genetically pure R.
rosea (and its extracts), they are not
necessarily the only pharmacologically active ingredients responsible for the
efficacy observed in clinical studies. In fact, precise identification of the
compounds responsible for the numerous health benefits of R. rosea remains to be confirmed.
R. rosea extracts
used in most human clinical studies were standardized to minimum 3 percent
rosavins and 0.8-1 percent salidroside because the naturally occurring ratio of
these compounds in R. rosea root is approximately 3:1.
Rhodiola rosea in Modern Medicine
Since 1969, R. rosea
has been included in official Russian medicine. The Pharmacological and
Pharmacopoeia Committee of the Soviet Ministry of Health recommended medicinal
use and industrial production of liquid R. rosea extract. In 1975, the Soviet Ministry of Health
approved and registered preparation No. 75/933/14 as a medicine and tonic,
allowing large-scale production under the name Rhodiola Extract Liquid, an
alcohol-based extract (40 percent ethyl alcohol). Medical and pharmacological
texts describe its use as a stimulant for asthenia (fatigue), for somatic and
infectious illnesses, in psychiatric and neurological conditions, and in healthy
individuals to relieve fatigue and to increase attention span, memory, and work
productivity. The common dose is 5-10 drops 2-3 times a day, 15-30 minutes
before eating for a period of 10-20 days. In psychiatric disorders with
fatigue, a starting dose of 10 drops 2-3 times a day is gradually increased up
to 30-40 drops for 1-2 months.
In Sweden, R. rosea
was recognized as an Herbal Medicinal Product in 1985 and has been described as
an antifatigue agent in the Textbook of Phytomedicine for Pharmacists.9 In the textbook of pharmacology for
dispenser training in Sweden, R. rosea is mentioned as a plant with a stimulant action. Also, the Pharmaceutical
Book (Lakemedelsboken 97/98) mentions R. rosea as one of the most commonly used psychostimulants in
the group of officially registered herbal medicinal products.11 In
Denmark, R. rosea is registered
as a medical product in the category of botanical drugs. Registered
preparations are extensively used in Sweden and other Scandinavian countries to
increase mental work capacity during stress, as a psychostimulant, and as a
general strengthener.
Pharmacological and Clinical Studies
The traditional use of R. rosea as a tonic in Siberian and Russian medicine
stimulated extensive research leading to identification of R. rosea as an adaptogen — a substance that nonspecifically
increases the resistance of an organism and does not disturb normal biological
parameters. Studies in cell cultures, animals, and humans have revealed
antifatigue, anti-stress, antihypoxic (protection against damaging effects of
oxygen deprivation), anticancer, antioxidant, immune enhancing and sexual
stimulating effects.2,18,24,38-40 Since the Russian and Bulgarian
literature is so extensive, this discussion will highlight seminal studies and
major reviews. The authors were fortunate to gain access to original reviews,
articles, and doctoral theses. This overview relies heavily on monographs and
peer-reviewed publications. The research data contained in these documents are
helpful for understanding recent human studies in normal and pathological
conditions.
Effects upon the Central Nervous System
The systematic study of the pharmacological effects of R.
rosea, begun in 1965, found that small and
medium doses had a simulating effect, such as lengthening the time mice swim
and remain on vertical perches to the limit of their abilities. In contrast,
larger doses were found to have more sedative effects. Small doses increased
the bioelectrical activity of the brain, presumably by direct effects on the
brainstem ascending and descending reticular formation.23-26,38,39,41
Further studies showed that medium range doses, unlike tranquilizers, enhanced
the development of conditioned avoidance reflexes in rats and facilitated
learning based on emotionally positive reinforcement.18,42-46 Overall,
in small and medium doses, R. rosea
stimulated norepinephrine (NE), dopamine (DA), serotonin (5-HT), and nicotinic
cholinergic effects in the central nervous system (CNS). It also enhanced the
effects of these neurotransmitters on the brain by increasing the permeability
of the blood brain barrier to precursors of DA and 5-HT.2,23,42,46-49
In comparing studies of R. rosea, Asian ginseng (Panax ginseng C.A. Mey., Araliaceae), meclofenoxate (centrophenoxine), piracetam, citicholine,
and other nootropics (substances that enhance cognition, protect the brain, and
have low toxicity and few side effects), Petkov and colleagues noted that all
of these agents enhance learning and memory in animal models and increase 5-HT
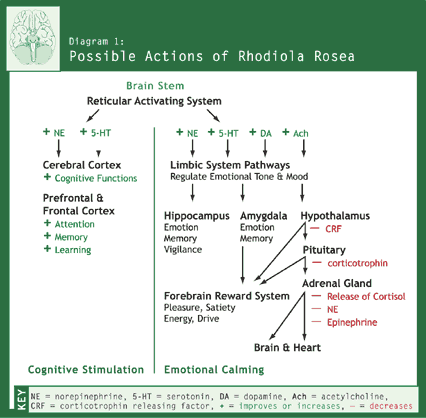
levels in the frontal cerebral cortex.46-50 Diagram 1 illustrates
the possible effects of R. rosea
on neurotransmitters in multiple neuronal pathways.51 Starting in
the brain stem, R. rosea promotes
release of NE, 5-HT, and DA in ascending pathways that activate the cerebral
cortex and the limbic system.2,49,50 Consequently, the cognitive
(thinking, analyzing, evaluating, calculating, and planning) functions of the
cerebral cortex and the attention, memory, and learning functions of the
prefrontal and frontal cortex are enhanced. Other neuronal systems also
contribute to the many aspects of memory: encoding, sorting, storage, and
retrieval. For example, the cholinergic system uses the neurotransmitter
acetylcholine (Ach) and contributes to memory function via pathways ascending
from the memory storage systems of the limbic system to various areas of the
cerebral cortex (memory retrieval). Agents that block Ach suppress the activity
of these ascending pathways and interfere with memory. R. rosea reverses this blockade.49,50 The deterioration of these systems with age results
in age-associated memory loss.52 R. rosea may prevent or ameliorate some age-related
dysfunction in these neuronal systems.
As an antioxidant,53-55 R. rosea may help protect the nervous system from oxidative
damage by free radicals. Stress interferes with memory functions and, over
time, causes deterioration in memory systems. In addition to enhancing
cognitive functions, learning, and memory by stimulating NE, DA, 5-HT, and Ach
neuronal systems, R. rosea may
exert positive effects on memory and cognition by improving resistance to
physical and emotional stress. Thus, the dual action of cognitive stimulation
and emotional calming creates benefits for both immediate cognitive and memory
performance and for the long-term preservation of brain functions.
The psychostimulant effects of R. rosea were studied in 53 healthy subjects and 412 patients
with neuroses and asthenic syndromes (of both functional and organic origin).56-58
Symptoms of asthenia (fatigue, decline in work capacity, trouble falling
asleep, poor appetite, irritability, and headaches) responded favorably to R.
rosea 50 mg three times a day. Treatment
durations ranged from 10 days to 4 months. The asthenic states included both
psychiatric and physical causes, for example, following influenza or other
illness. In an open study of 128 patients aged 17-55 years, R. rosea alleviated fatigue, irritability, distractibility,
headache, weakness and other vegetative symptoms in 64 percent of cases.57
Improvement was assessed by psychological testing and work productivity.
In 1869 Beard coined the term "neurasthenia" to include
various forms of nervous asthenia. Controversy over this term has centered on
the overlap of symptomatology and co-morbidity with other conditions (e.g.,
depression, neuroses, somatoform disorders, and chronic fatigue syndrome).
Although this diagnosis has fallen out of favor in the United States and no
longer appears in The Diagnostic and Statistical Manual of the American
Psychiatric Association (DSM-IV),59
it is still widely used throughout the world.60-63 Neurasthenia is
defined by the World Health Organization in the International
Classification of Diseases (ICD-10)64
as:
- either persistent and distressing feelings of exhaustion after minor mental effort, or
persistent and distressing feelings of fatigue after minor physical effort;
- accompanied
by one or more of the following symptoms: muscular aches or pains; dizziness;
tension headaches; sleep disturbance; inability to relax; and irritability;
- inability to recover through rest, relaxation, or enjoyment;
- does not occur in the presence of organic mental disorders, affective disorders or
panic, or generalized anxiety disorder.
In an open study 27 healthy students, physicians, and
scientists aged 19-46 years were given 10 drops of R. rosea tincture (equivalent to 100-150 mg R.
rosea extract) once or twice a day for 2-3
weeks, beginning several days before intense intellectual work, such as final
exams.58 The extract improved the amount and quality of work and in
all cases prevented asthenic decompensation (loss of work capacity due to
fatigue). A series of studies using a proofreading test showed that a one-time
dose of R. rosea did not
significantly increase the number of symbols corrected, but very significantly
decreased the percent of errors made, particularly over an 8-hour period.65,66
Positive results found in the studies of proofreading tests were based on 300
mg/day or more. In medical treatments, the usual doses are 200-600 mg/day. R.
rosea increased intellectual capacity
(particularly by improving perception and processing of information) to a
greater degree than an extract of eleuthero, formerly called Siberian ginseng (Eleutherococcus
senticosus Rupr. et Max., Araliaceae).18
The decrease in physical and mental performance of
physicians on prolonged night call is well known. Low dose (170 mg/day) R.
rosea extract was given to 56 young,
healthy physicians on night call.18 The effect was measured as total
mental performance calculated as "Fatigue Index." The tests reflected an
overall level of mental fatigue involving complex cognitive functions, such as
associative thinking, short-term memory, calculation, concentration, and speed
of audio-visual perception. These parameters were tested before and after night
duty during three periods of two weeks each in a double-blind crossover trial.
A statistically significant improvement in mental performance tests was
observed in the treatment group (R. rosea) during the first two-week period. However, at 6 weeks the effect
appeared to be lost. No side effects were reported. These results suggest that R.
rosea extract can reduce fatigue under
certain stressful conditions for some period of time. Possible reasons for the
loss of efficacy over time may be the low dose used, the crossover design, or
the overall length of night duty with increased fatigue by weeks 5 and 6.
Spasov and colleagues compared 100 mg/day R. rosea extract (SHR-5, Swedish Herbal Institute, Goteborg,
Sweden; standardized to 3 percent rosavin and 0.8 percent salidroside) with
placebo in a double-blind 20-day study of 60 Indian medical students studying
in Russia during their final exam period.38 Despite the low dosage,
investigators found significant improvements in general well-being, physical
fitness, mental fatigue, final exam grades, and coordination, but not in some
aspects of cognitive functioning in students taking R. rosea extract compared to placebo.
In a double-blind placebo-controlled study of 60 foreign
students at a Russian high school, administration of a R. rosea extract (660 mg/day of a preparation named Rodaxon)
resulted in an increase in physical (velergometric) work capacity,
coordination, kinesthetic sensitivity, and general well-being along with a
decrease in psychic fatigue and situational anxiety.39 Unfortunately,
this study provides no information on the amount of R. rosea in the Rodaxon preparation.
R. rosea was
beneficial in posttraumatic and vascular lesions of the brain. It was
especially effective in combination with piracetam for patients with marked
cognitive dysfunction.56 However, it did not reduce manic symptoms
and could worsen paranoid states. In one study of more clearly depressed
patients, R. rosea in combination
with tricyclic antidepressants (TCAs) produced significant improvement in the
majority of cases and decreased side effects of the TCAs.67
Ultimately, some of these patients were able to respond to R. rosea alone.
Antipsychotic medications used in large doses over many
years to treat schizophrenic patients sometimes affect the dopaminergic nerves
in the basal ganglia, the same nerves that are damaged in patients with
Parkinson's Disease. When these nerves are compromised, patients develop a
constellation of "Parkinsonian" symptoms, including stiffness, tremors,
bradykinesia (slowed movements), and others. Anticholinergic medications have
been used to relieve these symptoms when they are caused by antipsychotic
medication; however, they sometimes fail to help. In schizophrenic patients
whose anticholinergic medications had failed to relieve Parkinsonian symptoms, R.
rosea was found to be of benefit.56,68
R. rosea may affect
emotional tone by influencing neurotransmitter monoamine levels (NE, DA, 5-HT)
in nerve tracts involved in the regulation of mood, anxiety, and emotion in the
amygdala, hippocampus, hypothalamus, and midbrain. The stimulation of nicotinic
cholinergic activity in the emotional circuits of the limbic system (in the
temporal lobe) may also contribute to these effects. Alterations in monoamine
levels underlie this complex spectrum of psychotropic activity: stimulating,
tranquilizing, anti-stress, and antidepressant.
The authors have found that R. rosea can help patients with depressive syndromes, mental
and physical fatigue (secondary to psychiatric and medical conditions), memory
loss and cognitive dysfunction from a variety of causes, sexual dysfunction,
and menopausal-related disorders. Dr. Brown and Dr. Gerbarg have successfully
treated more than 150 individuals with R. rosea extract (3 percent rosavin and 1 percent
salidroside) and have supervised the treatment of more than 100 additional
cases (See Case Studies).
Effects on Physical Work
Capacity
A number of studies have shown that R. rosea increased physical work capacity and dramatically
shortened the recovery time between bouts of high-intensity exercise. These
studies included normal individuals exposed to maximal work on a bicycle
ergometer and Olympic-level cross country skiers and biathletes.69
In one study, 52 men (18-24 years of age) were given one dose of either 15
drops of R. rosea extract, 2 ml
eleuthero, or 1 ml of a 1 percent solution piridrol (a stimulating psychotropic
similar to methylphenidate). Fifteen drops of R. rosea extract is approximately equivalent to 150 mg of dry
encapsulated root extract standardized to 3 percent rosavin and 1 percent
salidroside. After 30 minutes, they pedaled an electric bicycle ergometer to
produce a precise amount of work-induced baseline fatigue. After a 5-minute
rest, they performed further work to determine the maximal duration of work
they could accomplish at a specific intensity. During the second period of
work, R. rosea drops, eleuthero
extract, and piridrol increased work capacity by 9 percent, 6 percent, and 6
percent respectively (p<0.04) compared to placebo controls. Recovery was
defined by the time of normalization of heart rate and arterial pressure.
During the recovery period, at 10 minutes, the pulse slowed by a factor of 2.5
(67 beats per minute) in the R. rosea group versus 1.9 (87 beats per minute) in the control group. During
the 3-day total recovery period, subjects given piridrol complained of
insomnia, excitability, and irritability; whereas those given R.
rosea had no adverse side effects and no
complaints.
Endurance is the capacity to maintain work despite fatigue.
Forty-two master level competitive skiers (20-25 years of age) took either R.
rosea extract or placebo 30-60 minutes before training races (30
km) and a biathlon (20 km race on skis carrying a rifle and shooting targets at
stops). Athletes given R. rosea
had statistically significant increased shooting accuracy, less arm tremor and
better coordination. Thirty minutes after work performance, the heart rate in
the R. rosea group was 104-106
percent of baseline, versus 128.7 percent in the placebo group (p<0.02). R.
rosea improved recovery time, strength,
endurance, cardiovascular measures, and coordination.69
Adaptogens differ from other stimulants during forced, exhaustive
muscular work. With classical stimulants the initial increase in work-capacity
is followed by a period of substantially decreased (markedly below average)
work-capacity. Repeated use of CNS stimulants depletes brain catecholamines and
decreases conditioned reflexes. In contrast, with extracts of R. rosea, the initial increase in work-capacity is followed
by a lesser diminution, such that the work-capacity continues to be above
average.70
Animal studies suggest mechanisms that may be involved in
these effects. R. rosea increased
essential energy metabolites, adenosine triphosphate (ATP), and creatine
phosphate in the muscle and brain mitochondria in mice made to swim to their
limit.71 It may also enhance the ammonia reassimilation and energy
metabolism of the cell by increasing ATP, ribonucleic acid (RNA), protein, and
amino acid synthesis.72 In animal studies, R. rosea increased metabolism of fats twice as much as
eleuthero73 and improved energy metabolism in the brain during
intensive muscular workloads.74
Adaptogenic, Anti-Stress, and Neuroendocrine Effects
In their classic 1968 paper, Soviet pharmacologists Brekhman
and Dardymov surveyed the literature on 189 medicinal plants and identified
five (including R. rosea) that met the
three defining criteria for an adaptogen:75
- An adaptogen should be innocuous and cause minimal disturbance of the normal
physiological functions of an organism;
- The action of an adaptogen should be nonspecific (i.e., it should increase
resistance to adverse influences of a wide range of harmful factors of
physical, chemical, and biological nature);
- An adaptogen may possess normalizing action irrespective of the direction of the
preceding pathological changes (i.e., if a body parameter is high, the
adaptogen brings it down towards normal; if a parameter is low, the adaptogen
brings it up towards normal).
The forced swimming test, used by Russian scientists to
measure nonspecific resistance to stress, was later named after Porsolt who
assigned specific parameters such as water temperature and the dimensions of
the glass cylinder in which a mouse or rat was forced to swim to exhaustion
(about 15 minutes). After an initial period of vigorous activity, the rodent
adopts a characteristic immobile posture, making only the minimal movements
necessary to stay afloat.76 The validity of the Porsolt swim test
and its relationship to depression have been discussed extensively77,78
and it subsequently became a screening test for antidepressant agents by
pharmaceutical companies. Although different laboratories have made minor
technical modifications, the fundamentals of the test remain the same.
Adaptogens and antidepressants increase the amount of time the animal is able
to keep swimming actively.75 Panossian and colleagues propose to
update the definition of adaptogen by highlighting more specific biochemical
actions as metabolic regulators.70 The wide range of medical
benefits and physiological actions may be based on the effects of adaptogens on
regulatory systems found in many organs and tissues (e.g., immune, hormonal,
CNS, cardiovascular, muscular, etc.). They hypothesize that adaptogens reduce
damage from stressors by altering the reactivity of the organism's defense
system, including the hypothalamic pituitary axis (HPA) and the efferent
sympatho-adrenal system (SAS).70
A recent study showed that R. rosea and eleuthero protected the embryos of freshwater
snails (Lymnaea stagnalis)
from a variety of environmental stressors.79
Enhancement in resistance was studied by applying phyto-adaptogen extracts for
a period of 20 hours to 3-day old L. stagnalis larvae. Subsequently the larvae were exposed to the
following highly toxic environmental stressors: a physical stress (heat shock:
43 degrees C for 4 minutes); an oxidative stress (superoxide radicals induced
by menadione 600 microM for 2 hours); and heavy metal-induced stress (copper 50
microM for 1 hour or cadmium 20 microM for 1 hour). Both eleuthero and R.
rosea strongly protected snail embryos from
lethal heat shock, from the adverse effects of menadione-induced superoxide
radicals, and from toxic exposure to heavy metals (copper and cadmium).
Although the degree to which resistance was enhanced depended on the type of
stressor applied, these results confirm the definition of phyto-adaptogens as
being universal enhancers of non-specific resistance against different kinds of
stress conditions. The mechanisms of nonspecific resistance are not entirely
clear, but probably involve improvements in cellular energy metabolism, based
in part on ATP (as discussed above).
In higher animals and humans, nonspecific resistance may
also be enhanced by improvements in the neurological mechanisms of dealing with
stress (catecholamines, serotonin, and endorphins). The serotonin system is
necessary for the stress response reaction, adaptation to new environmental
conditions, and tolerance of hypoxia. Numerous stressors decrease serotonin in
the hypothalamus. Theoretically, the ability of R. rosea to increase the nonspecific resistance of animals
may be related to its capacity to increase serotonin in the hypothalamus and
midbrain. Additional research showed that an intact hypothalamic pituitary
adrenal axis and participation of the gonads and thymus were necessary for this
anti-stress effect.2 Furthermore, R. rosea reduces
the activation of several components of the stress response system. For
example, it modestly increased serum beta-endorphins that protected rats
against subsequent stress-induced excess endorphin elevation.80 In
addition, R. rosea moderates the
release of opioid peptides that occurs as part of the pituitary adrenal axis
response to stress. This reduced release protects against sudden excess opioid
and catecholamine (NE and DA) levels (which interfere with normal brain
functions and can lead to heart damage), while allowing a more moderate release
that increases stress tolerance without damaging the central nervous system or
the cardiovascular system (see Diagram 2). R. rosea extracts also protect the brain and heart by
reducing the secretion of corticotrophin releasing factor (CRF) under stress.80,81

Endocrine and Reproductive Effects
Neuroendocrine animal studies showed that R. rosea, like other adaptogens, enhanced thyroid function
without causing hyperthyroidism.81 In addition, the thymus gland
functioned better and was protected from the involution that occurs with aging.
The adrenal glands functioned with better reserve and without the kind of
hypertrophy caused by other psychostimulants.
Egg maturation was enhanced in rats and an anabolic effect
in males (increased muscle building and gonad strengthening similar to effects
of low-dose testosterone) was observed in a number of species. Administration
of rhodosin (extract of R. rosea for
intravenous, intramuscular, or peritoneal injection) to sexually mature female mice over a period of 4
weeks prolonged menstruation from 1.3 days (control) to 2.8 days (rhodosin
treated), reduced the resting period from 3.8 days (control) to 2.2 days
(rhodosin treated), and increased the relative number of estrus days from 29
percent to 56 percent. In the majority of rhodosin treated animals, the number
of growing follicles, the oocyte volumes, the accumulation of RNA in oocyte
cytoplasm, the proliferation of the lining and glandular cells of the uterine
horns, and the preparation of uterine mucosa for fertilization all increased.
In sexually mature mice, rhodosin increased the mean weight of the uterine
horns from 39.6+4.11 mg to 59.5+1.59 mg and the mean weight of
the ovaries from 6.4+0.65 mg to 9.1+0.45 mg. However, the
administration of rhodosin to sexually immature female white mice for 3 weeks
did not affect sexual maturation, the onset of estrus, the weight of ovaries or
uterine horns, or the maturation of follicles. Thus, it is probable that the
estrogenic effects of R. rosea
preparations depend upon a specific hormonal milieu.82,83
These pre-clinical investigations led to a study of R.
rosea extract in women suffering from
amenorrhea (loss of menstrual cycles). Forty women with amenorrhea were given R.
rosea (either 100 mg R. rosea extract orally twice a day for 2 weeks, or 1 ml
rhodosin intramuscularly for 10 days). In some subjects the treatment cycle was
repeated 2-4 times. Normal menses were restored in 25 women, 11 of whom became
pregnant. In those with normal menses, the mean length of the uterine cavity
increased from 5.5 cm to 7.0 cm (normal) after R. rosea treatment.82,83 One of the authors (Dr.
Brown) has treated in his practice several women who had failed to conceive
with standard fertility drugs, and who become pregnant within several months of
beginning R. rosea extract. These
preliminary clinical observations warrant controlled follow-up clinical trials.
Using the in vitro estrogen
receptor competition assay, Patricia Eagon, Ph.D. (personal communication,
December 2001) recently found that R. rosea extract showed strong estrogen binding properties
that require further characterization.
In an open study, 26 out of 35 men with erectile dysfunction
and/or premature ejaculation (of 1-20 years duration) responded to R. rosea
(150-200 mg/day for 3 months) with substantially improved sexual function,
normalization of prostatic fluid, and an increase in 17-ketosteroids in urine.56,69
Cardioprotective Effects
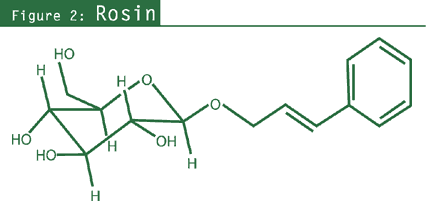
Cardioprotective effects of R. rosea include: prevention of stress-induced cardiac
damage,80,81,84 decreased myocardial catecholamines and cyclic
adenosine monophosphate (cAMP) levels; and reduced adrenal catecholamine
release80,81 (see Figure 2). Furthermore, R. rosea activation of mu-opiate receptors in heart muscle
prevented reperfusion arrhythmias in animal hearts. This effect could be
blocked by naloxone injection (known to inhibit mu-opiate receptors), thus
confirming that the anti-arrhythmic effect of R. rosea is associated with the mu-opiate receptors in
myocardial (heart) muscle.84
In a series of joint Swedish and Russian double-blind,
randomized placebo-controlled studies,85 10 healthy but sedentary
men (ages 20-31 years) were evaluated. Twenty percent of the subjects had average
physical work capacity as measured by Power Work Capacity (PWC-170) and 80
percent had below-average PWC-170, indicating a low level of physical training
(PWC-170 is a calculation based on the amount of work performed by a man if his
heart rate reaches 170 beats per minute, bpm). A sequence of complex 1- to
7-day trials compared the effects of an adaptogen formula, a mixture of mono-
and polyphenolic adaptogens (MMPA). Each tablet contained the following
ingredients: 3 mg rhodioloside from R. rosea root extract, 50 mg; 3 mg total sum of isofraxidine-, syringine-, and
syringaresinoie-glycosides from eleuthero root extract, 100 mg; and 4 mg
schizandrine and gamma-schizandrine from schisandra (Schisandra
chinensis (Turcz.) Baill., Lamiaceae) fruit
extract, 150 mg.
During the 7-day adaptogen trial, subjects were given 3
capsules (containing a total of 150 mg R. rosea) twice a day on days 1-3; 4 capsules (200 mg R. rosea) twice a day on days 4-6, and 4 capsules once on day
7. The mean increase in physical work capacity was 28 percent with dosed
physical loads in subjects treated with the adaptogen formula. Thus, sedentary
subjects given the adaptogen were able to perform in the lower level of trained
athletes without any exercise training. Their heart rate variability and
inotropic (strength of heart muscle contractility) functions improved.
Both the sympathetic and parasympathetic inputs to the heart
were enhanced such that the heart showed increased reserves under stress of
greater intensity. The autonomic nervous system controls automatic or
involuntary functions of the body. It has two components: the sympathetic and
the parasympathetic nerves (see Diagram 2). The sympathetic nervous system is
the "fight-or-flight" system that helps the organism respond to stress (e.g.,
by increasing heart rate, respiratory rate, and muscle tone). The
parasympathetic nervous system conserves and restores energy (e.g., by slowing
the heart rate, respiratory rate, and metabolism). By enhancing the functions
of the sympathetic and parasympathetic systems, R. rosea enables the organism to put out more energy during
stress while at the same time maintaining higher energy reserves. One of the
challenges presented by research on a multi-ingredient formula is that it is
not usually possible to attribute the results to the activity of any one single
herbal component. However, the results of this study are consistent with
results of other research conducted solely on R. rosea monopreparations.
Antioxidant and Anti-carcinogenic Effects
R. rosea is rich in
phenolic compounds, known to have strong antioxidant properties.53,86 Animal studies have shown that R.
rosea decreases toxicity from
cyclophosphamide, rubomycin, and adriamycin (anti-cancer drugs), while it
enhances their anticarcinogenic effects.87-89 Udintsev and Schakhov
studied the effect of R. rosea
root extract (RRRE), a tincture manufactured according to the Russian
Pharmacopoeia standards (minimum 0.8 percent salidroside and 3 percent
rosavin), on tumor cells (transplanted into mice) and normal bone marrow cells
in two mouse cancer models.90 One group of mice with Ehrlich ascites
tumor (EAT) and another group with Lewis lung carcinoma (3LL) were first
treated with 100 mg/kg cyclophosphamide (a chemotherapy agent) that suppressed tumor
growth to 31-39 percent and limited 3LL metastases to 18 percent, while also
reducing the number of normal bone marrow cells, leucocytes, and
myelokariocytes, to 40-50 percent and 20-25 percent of normal, respectively. In
comparison, RRRE, 0.5 mg/kg/day given orally 2-8 days after tumors had been
transplanted, suppressed growth of both tumors by 19-27 percent and 3LL
metastases 16 percent. However, in contrast to cyclophosphamide, RRRE caused no
reduction in normal bone marrow cells. In animals given both RRRE and
cyclophosphamide, the RRRE increased the antimetastatic effect of
cyclophosphamide by 36 percent (p<0.05). RRRE also increased the number of
leukocytes by 30 percent and myelokariocytes by 16-18 percent.
In another mouse tumor model, Udintsev and colleagues showed
that RRRE (minimum 0.8 percent salidroside and 3 percent rosavin) increased the
antitumor effect of the drug adriamycin while substantially reducing its liver
toxicity.89 Many chemotherapy agents are hematotoxic (reduce the
number of normal blood cell precursors in bone marrow) or hepatotoxic (cause
damage to the liver). These serious side effects were significantly ameliorated
by RRRE. Thus, the research suggests that RRRE can both enhance tumor
inhibition by chemotherapeutic drugs while alleviating dangerous side effects.
Substances that reduce the incidence of chromosomal
aberrations are termed antimutagenic. Salikhova and colleagues found that in
mice injected with cyclophosphamide, RRRE (minimum 0.8 percent salidroside and
3 percent rosavin) had antimutagenic effects.91 Compared to placebo
controls, RRRE reduced the development of chromosomal aberrations by 50 percent
and reduced the incidence of cells with micronuclei by more than 50 percent.
RRRE also increased indices of DNA repair in bone marrow cells after exposure
to the mutagen N-nitroso-N-methylurea (NMU).91
In a small pilot study of 12 patients with superficial
bladder carcinoma (TIG1-2), treatment with RRRE (minimum 0.8 percent
salidroside and 3 percent rosavin) improved parameters of leukocyte integrines
and T-cell immunity.92 The average frequency of relapse was reduced,
but did not reach statistical significance. Larger placebo-controlled studies
of R. rosea extracts to augment tumor
inhibition and reduce toxic effects of chemotherapy agents are needed.
Toxicity, Side Effects, and Contraindications
R. rosea has a very
low level of toxicity. In rat toxicity studies, the LD50 (lethal dose at which
50 percent of animals die) was calculated to be 28.6 ml/kg, approximately 3,360
mg/kg.25 The equivalent dosage in a 70 kg man would be about 235 gm
or 235,000 mg. Since the usual clinical doses are 200-600 mg/day, there is a
huge margin of safety.87
Overall, R. rosea has
very few side effects. Most users find that it improves their mood, energy
level, and mental clarity. Some individuals, particularly those who tend to be
anxious, may feel overly activated, jittery, or agitated. If this occurs, then
a smaller dose with very gradual increases may be needed. R. rosea should be taken early in the day because it can
interfere with sleep or cause vivid dreams (not nightmares) during the first
few weeks. It is contraindicated in excited states. Because R. rosea has an activating antidepressant effect, it should
not be used in individuals with bipolar disorder who are vulnerable to becoming
manic when given antidepressants or stimulants. Until this has been further
studied, the authors advise caution in patients with bipolar spectrum
disorders. The herb does not appear to interact with other medications, though
it may have additive effects with other stimulants. It is best absorbed when
taken on an empty stomach 30 minutes before breakfast and lunch. As with any
herbal preparation, patients should inform their primary healthcare practitioner
when taking R. rosea.
Rhodiola in the Future
More scientific research is needed to confirm the preventive
and curative benefits of R. rosea.
Controlled studies are warranted to explore its use in antidepressant
augmentation, disorders of memory and cognition, attention deficit disorder,
traumatic brain injury, Parkinson's disease, protection against arrhythmias,
sports performance, aviation and space medicine (enhancing physical and mental
performance while reducing stress reactions), endocrine disorders (infertility,
premenstrual disorder, menopause), sexual dysfunction, disorders of the stress
response system (fibromyalgia, chronic fatigue syndrome, and post traumatic
stress disorder), and enhancement of chemotherapy/radiation with amelioration
of toxicity.
In the course of evolution, R. rosea has adapted to the harsh conditions of high altitude
(extreme cold, low oxygen, little rainfall, and intense irradiation from the
sun) by producing a group of powerful protective compounds that have diverse
beneficial effects in animals and humans. One is struck by the versatility of R.
rosea, from its description in Greek
medicine, 2000 years ago to its use by 20th century cosmonauts. It is time for
modern research, using controlled clinical trials, to develop the potential
medical applications of this unique phyto-adaptogen.
Richard P. Brown, M.D., is Associate Clinical Professor
of Psychiatry at Columbia University College of Physicians and Surgeons in New
York City. He received his M.D. in 1977 from Columbia University College of
Physicians and Surgeons in New York. Dr. Brown completed his Residency in
Psychiatry and a Fellowship in Psychobiology and Psychopharmacology at New York
Hospital. Dr. Brown is the recipient of numerous awards, including a
Mead-Johnson Neuropsychopharmacology Travel Fellowship, a Mallinckrodt Scholar
award, and a Fellowship in Neurosciences and Brain Imaging from the Dana
Foundation. He has had a longstanding interest in herbal and complementary
medicine, especially as relevant to psychiatry. In 2000, he co-authored the
book, Stop Depression Now, which
presents a holistic approach to the treatment of depression, including SAM-e.
Since 1999, Dr. Brown has taught a full day course on Herbs and Nutrients in
Psychiatry at the annual meetings of the American Psychiatric Association. Dr.
Brown has no financial interest in Rhodiola
rosea.
Patricia L. Gerbarg, M.D., is Assistant Clinical
Professor in Psychiatry at New York Medical Center. She graduated from Harvard
Medical School in 1975 and completed her Psychiatry Residency at Beth Israel
Hospital in Boston in 1979. She finished psychoanalytic training at the Boston
Psychoanalytic Society and Institute in 1992 and has maintained a private
psychiatric practice for 23 years. Dr. Gerbarg has taught and lectured on a
range of topics in psychiatry and psychoanalysis. Over the past eight years she
has been increasingly involved in research and writing about alternative and
complementary medicine in psychiatry and has co-authored numerous articles and
book chapters with Dr. Brown. Dr. Gerbarg has no financial interest in Rhodiola rosea.
Zakir Ramazanov, Ph.D., D.S., is Professor of
Biochemistry at Las Palmas Technological Institute, Spain. In 1978 he received
a bachelor's degree with a double major in biochemistry and plant physiology
from North Caucasian State University and in 1981 a Ph.D. in Plant Physiology
and Biochemistry from the Soviet Academy of Sciences. He has served as Senior
Scientist and Chief of the Department of Biotechnology at the Soviet Academy of
Science and as Chairman of Algal Biotechnology Development. In 1991 he accepted
a research fellowship at Louisiana State University. The recipient of numerous
research grants, Dr. Ramazanov is known for his work in space biology, the
cultivation of photosynthetic organisms in space stations, and the development
of marine natural products from sea vegetables. He has published more than 140
scientific studies and co-authored two books: Arctic
Root (Rhodiola rosea) — The powerful new Ginseng Alternative (1998)
and Effective Natural Stress and Weight
Management Using Rhodiola Rosea and Rhododendron Caucasicum (1999).
Dr. Ramazanov is President and CEO of National Biosciences Corporation,
Chester, NY.
Acknowledgement: The authors are grateful to Dr. Bertalan
Galambosi from Agrifood Research Finland, Ecological Production, of Mikkeli,
Finland, for providing the photographs of Rhodiola
rosea.
References
- Engler A. Syllabus der Pflanzenfamilien. Vol. 2.
Berlin, Germany: Borntraeger; 1964. p. 199-200.
- Saratikov
AS, Krasnov EA. Rhodiola rosea is a
valuable medicinal plant (Golden Root).
Tomsk, Russia: Tomsk State University Press; 1987.
- Mell CD.
Dyes, tannins, perfumes, and medicines from Rhodiola
rosea. Textile Colorist
1938;60(715):483-4.
- Linnaeus C. Materia Medica. Liber I. De Plantis.
Stockholm, Sweden: Lars Salvius; 1749. p. 168.
- Linnaeus C. Örtabok. Stockholm, Sweden: Almquist and
Wiksell; 1725. p. 127.
- Linnaeus C. Plants of Lapland. Uppsala, Sweden: The
Royal Science Academy’s documents; 1754.
p. 182-7.
- Linnaeus C. Flora Oeconomica eller Hushålls-Nyttan af de
i Swerige, Wildt wäxande Örter. Stockholm, Sweden: Lars Salvii; 1748. p.
399.
- Linnaeus C,
Tonning H. Norwegian Rarities.
Uppsala, Sweden: Johan Edman;1768.
p. 3-19.
- Sandberg F,
Bohlin L. Fytoterapi: växbaserade
läkemedel [Remedies based on herbs].
Stockholm, Sweden: Hälsokostrådets förlag AB; 1993. p. 131.
- Commission
Nationale de la Pharmacopée Française. Pharmacopée
Française. IX ed. Table alphabétique revisée des drogues végétales. Paris,
France: French Agence du Médicament–Direction des French Laboratoires et des
Contrôles, Unité Pharmacopée; 1974. p. II A214-101.
- Sandberg F. Herbal Remedies and Herb Magic.
Stockholm, Sweden: Det Bästa; 1998. p. 223.
- Mashkovskij
MD. Doctor’s manual: medical drugs.
12th ed. Vol. 1. Moscow, Russia: Meditzina; 1976. p. 161-5.
- Khaidaev Z,
Menshikova TA. Medicinal Plants in
Mongolian Medicine. Ulan-Bator, Mongolia; 1978.
- Magnusson B. Fägringar: Växter som berör oss (Beauty: herbs that touch us). Östersund,
Sweden: Berndtssons; 1992. p. 66-7.
- Hoppe H. Drogen kunde. Band 1, Angiosperm
8.Berlin, Germany: Walter de Gruyter; 1975. p. 986-7.
- Nörr H.
Phytochemical and pharmacological investigation of the adaptogens: Eleutherococcus senticocus, Ocimum sanctum,
Codonopsis pilosula, Rhodiola crenulata [dissertation]. Munich, Germany:
Faculty of Chemistry and Pharmacy, Ludwig-Maximilians-Universität München;
1993.
- Krylov GV. Herbs for Life. Novosibirsk, Russia:
Academic Press; 1969. p. 264.
- Darbinyan V,
Kteyan A, Panossian A, Gabrielian E, Wikman G, Wagner H. Rhodiola rosea in stress induced fatigue: a double blind cross-over
study of a standardized extract SHR-5 with a repeated low-dose regimen on the
mental performance of healthy physicians during night duty. Phytomedicine 2000;7(5):365-71.
- Komarov VL,
editor. Flora of the USSR. Volume IX,
Rosales and Sarraceniales. Genus 698: Rhodiola
L. Leningrad, Russia: The USSR Academy of Sciences: 1939. Translation:
Jerusalem, Israel: Israel Program for Scientific Translation; 1971. p. 20-36.
- Hegi G,
editor. Illustrierte Flora von
Mitteleuropa. Vol.IV/2, Liefering 2/3. Hamburg/Berlin, Germany; P. Posey:
1963. p. 99-102.
- Ohba H. A
revision of the eastern Himalayan species of the subgenus Rhodiola of the genus Sedum.
In: Ohashi H, editor. Flora of Eastern
Himalaya, 3rd report. Tokyo, Japan: University of Tokyo Press; 1975. p.
283-362.
- Saratikov AS,
Krasnov EA. Chapter I: Chemical composition of Rhodiola rosea. In: Saratikov AS, Krasnov EA. Rhodiola rosea is a valuable medicinal plant (Golden Root). Tomsk, Russia: Tomsk State University; 1987. p. 3-39.
- Kurkin VA,
Zapesochnaya GG. Chemical composition and pharmacological properties of Rhodiola rosea. Chemical and Pharmaceutical Journal (Moscow) 1986;20(10):1231-44.
- Saratikov AS.
Golden Root (Rhodiola rosea). Tomsk,
Russia: Tomsk State University Press; 1974.
- Kurkin VA,
Zapesochnaya GG. Chemical composition and pharmacological characteristics of Rhodiola rosea [review]. Journal of Medicinal Plants, Russian Academy
of Science, Moscow 1985;1231-445.
- Saratikov AS,
Krasnov EA, Khnikina LA, Duvidson LM. Isolation and chemical analysis of
individual biologically active constituents of Rhodiola rosea. Proceedings of the Siberian Academy of Sciences. Biology 1967;1:54-60.
- The Russian
Federation Ministry of Health and Medical Industry. Russian National Pharmacopoeia. Pharmacopoeia article: PA
42-2126-83, liquid extract of Rhodiola rosea root and rhizome. Moscow, Russia:
The Russian Federation Ministry of Health and Medical Industry; 1983.
- Dubichev AG,
Kurkin BA, Zapesochnaya GG, Vornotzov ED. Study of Rhodiola rosea root chemical composition using HPLC. Cemico-Parmaceutical Journal
1991;2:188-93.
- Ganzera M,
Yayla Y, Khan IA. Analysis of the marker compounds of Rhodiola rosea L. (golden root) by reversed phase high performance
liquid chromatography. Chem Pharm Bull
(Tokyo) 2001;49(4):465-7.
- 30. Zhang S, Wang
J, Zhang H. Chemical constituents of Tibetan medicinal herb Rhodiola kirilowii (Reg.). Gansu Chung Kuo Chung Yao Tsa Chih
1991;16(8):483, 512.
- 31. Wang S, Wang
FP. Studies on the chemical components of Rhodiola
crenulata. Yao Hsueh Hsueh Pao
1992;27(2):117-20.
- 32. Wang S, You
XT, Wang FP. HPLC determination of salidroside in the roots of Rhodiola genus plants. Yao Hsueh Hsueh Pao 1992;27(11):849-52.
- 33. Bridel M,
Beguin C. Isolation of rutoside, asparagines and a new glycoside, hydrolysable
by emulsion, salidroside from Salix
triandra L. Séances Acad Sci 1926;183:321-3.
- 34. Shi L, Ma Y,
Cai Z. Quantitative determination of salidroside and specnuezhenide in the
fruits of Ligustrum lucidum by high
performance liquid chromatography. Biomed
Chromatogr 1998;12(1):27-30.
- 35. Thieme H. On
the identity of glucoside rhodioloside and salidroside. Pharmazie 1969;24(2):118-9.
- 36. Thieme H,
Walewska E, Winkler HJ. Isolation of salidroside from leaves of Rhododendron ponticum x catawbiense. Pharmazie 1969;24(12):783.
- 37. The Russian
Federation Ministry of Health and Medical Industry. Russian National Pharmacopoeia. Moscow, Russia: The Russian
Federation Ministry of Health and Medical Industry; 1989.
- 38. Spasov AA,
Wikman GK, Mandrikov VB, Mironova IA, Neumoin VV. A double-blind,
placebo-controlled pilot study of the stimulating and adaptogenic effect of Rhodiola rosea SHR-5 extract on the
fatigue of students caused by stress during an examination period with a
repeated low-dose regimen. Phytomedicine
2000;7(2):85-9.
- 39. Spasov AA,
Mandrikov VB, Mironova IA. The effect of the preparation rhodiosin on the
psychophysiological and physical adaptation of students to an academic load. Eksp Klin Farmakol 2000;63(1):76-8.
- 40. Furmanowa M,
Oledzka H, Michalska M, Sokolnicka I, Radomska D. Chapter XXIII Rhodiola rosea L. (Roseroot): In vitro regeneration and the biological
activity of roots. Vol. 33. In: Bajaj YPS, editor. Biotechnology in Agriculture and Forestry. Vol. 33. Medicinal and aromatic plants. VIII. Berlin and Heidelberg, Germany:
Springer-Verlag; 1995. p. 412-26.
- 41. Saratikov AS,
Krasnov EA. Chapter VII: Adaptogenic properties of Rhodiola rosea. In: Saratikov AS, Krasnov, editors. Rhodiola rosea is a valuable medicinal plant
(Golden Root). Tomsk, Russia:
Tomsk State University Press; 1987. p. 194-215.
- 42. Petkov VD,
Stancheva SL, Tocuschieva L, Petkov VV. Changes in brain biogenic monoamines
induced by the nootropic drugs adafenoxate and meclofenoxate and by citicholine
(experiments on rats). Gen Pharmacol
1990;21(1):71-5.
- 43. Baranov VB.
Experimental trials of herbal adaptogen effect on the quality of operation
activity, mental and professional work capacity. Contract 93-11-615 Stage 2
Phase I. Moscow, Russia: Russian Federation Ministry of Health Institute of
Medical and Biological Problems; 1994.
- 44. Komar VV, Kit
SM, Sischuk LV, Sischuk VM. Effect of Rhodiola
rosea on the human mental activity. Pharmaceutical
J 1981;36(4):62-4.
- 45. Stancheva SL,
Mosharrof A. Effect of the extract of Rhodiola
rosea L. on the content of the brain biogenic monoamines. Medecine Physiologie Comptes Rendus de
l’Académie Bulgare des Sciences 1987;40(6):85-7.
- 46. Lazarova MB,
Petkov VD, Markovska VL, Petkov VV, Mosharrof A. Effects of meclofenoxate and
extr. Rhodiolae rosea L. on
electroconvulsive shock-impaired learning and memory in rats. Methods Find Exp Clin Pharmacol
1986;8(9):547-52.
- 47. Petkov VD,
Yonkov D, Mosharoff A, Kambourova T, Alova L, Petkov VV, et al. Effects of
alcohol aqueous extract from Rhodiola
rosea L. roots on learning and memory. Acta
Physiol Pharmacol Bulg 1986;12(1):3-16.
- 48. Saratikov A,
Marina TF, Fisanova LL. Effect of golden root extract on processes of serotonin
synthesis in CNS. Journal of Biological
Sciences 1978;6:142.
- 49. Marina TF,
Alekseeva LP. Effect of Rhodiola rosea
extract on electroencephalograms in rabbit. In. Saratikov AS, editor. Stimulants of the Central Nervous System.
Tomsk, Russia: Tomsk State University Press; 1968. p. 22-6.
- 50. Marina TF.
Effect of Rhodiola rosea extract on
bioelectrical activity of the cerebral cortex isolated to a different extent from
the brain. In. Saratikov AS, editor. Stimulants
of the Central Nervous System. Tomsk, Russia: Tomsk State University Press;
1968. p. 27-31.
- 51. Brown RP,
Gerbarg PG, Muskin PR. Alternative Therapies in Psychiatry. In: Tasman A,
Lieberman J, Kay J, editors. Psychiatry. 2nd
ed [in press]. West Sussex, England: Wiley & Sons, Ltd.; 2002.
- 52. Lupien SJ, de
Leon M, de Santi S, Convit A, Tarshish C, Nair NP, et al. Cortisol levels
during human aging predict hippocampal atrophy and memory deficits. Nat Neurosci 1998;1(1):69-73.
- 53. Furmanowa M,
Skopinska-Rozewska E, Rogala E, Malgorzata H. Rhodiola rosea in vitro culture: phytochemical analysis and
antioxidant action. Acta Societis
Botanicorum Poloniae 1998;76(1):69-73.
- 54. Durany N,
Munch G, Michel T, Riederer P. Investigations on oxidative stress and
therapeutical implications in dementia. Eur
Arch Psychiatry Clin Neurosci 1999;249 Suppl 3:68-73.
- 55. Joseph JA,
Shukitt-Hale B, Denisova NA, Bielinski D, Martin A, McEwen JJ, et al. Reversals
of age-related declines in neuronal signal transduction, cognitive, and motor
behavioral deficits with blueberry, spinach, or strawberry dietary
supplementation. J Neurosci
1999;19(18):8114-21.
- 56. Saratikov AS,
Krasnov EA. Chapter VIII: Clinical studies of Rhodiola. In: Saratikov AS, Krasnov EA, editors. Rhodiola rosea is a valuable medicinal plant
(Golden Root). Tomsk, Russia:
Tomsk State University Press; 1987. p. 216-27.
- 57. Krasik ED,
Morozova ES, Petrova KP, Ragulina GA, Shemetova LA, Shuvaev VP. Therapy of
asthenic conditions: clinical perspectives of application of Rhodiola rosea extract (golden root).
In. Proceedings Modern problems in
psycho-pharmacology. Kemerovo-city, Russia: Siberian Branch of Russian
Academy of Sciences: 1970. p. 298-330.
- 58. Krasik ED,
Petrova KP, Rogulina GA, Shemetova LYa, Shuvayeva. New data on the therapy of
asthenic conditions (clinical prospects for the use of Rhodiola extract). Proceedings of All-Russia Conference: Urgent
Problems in Psychopharmacology 1970 May 26-29. Sverdlovsk, Russia: Sverdlovsk
Press; 1970. p. 215-7.
- 59. American
Psychiatric Association. Diagnostic and
Statistical Manual of Mental Disorders. 4th ed (DSM-IV). Washington, DC:
American Psychiatric Association; 1994.
- 60. Hickie I,
Davenport T, Issakidis C, Andrews G. Neurasthenia: prevalence, disability, and
healthcare characteristics in the Australian community. Br J Psychiatry 2002;181:56-61.
- 61. Merikangas K,
Angst J. Neurasthenia in longitudinal cohort study of young adults. Psychological Medicine
1994;24(4):1013-24.
- 62. Lin TY.
Neurasthenia revisited: its place in modern psychiatry. Psychiatric Annals 1992;22(4):173-87.
- 63. Zheng YP, Lin
KM, Takeuchi D, Kurasaki KS, Wang Y, Cheung F. An epidemiological study of
neurasthenia in Chinese-Americans in Los Angeles. Comprehensive Psychiatry 1997;38(5):249-59.
- 64. U.S.
Department of Health and Human Services. The
International Classification of Diseases. 10th Revision (ICD-10).
Washington, DC: U.S. Department of Health and Human Services; 1992.
- 65. Zatova MI,
Krilov GV, Saratikov AS. Golden root: new stimulative and adaptogenic drug. Proceedings of Siberian Academy of Sciences.
Biological and Medical Sciences. 1965;2:111-9.
- 66. Saratikov AS.
Screening for natural central nervous system stimulants. In: Saratikov AS,
editor. Stimulants of the Central Nervous
System. Vol. 1. Tomsk, Russia: Tomsk State University Press; 1966 p. 3-23.
- 67. Brichenko VS,
Kupriyanova IE, Skorokhova TF. The use of herbal adaptogens with tricyclic
antidepressants in patients with psychogenic depression. In: Saratikov AS,
editor. Modern Problems of Pharmacology
and Search for New Medicines. Tomsk, Russia: Tomsk State University Press;
1986. p. 58-60.
- 68. Mattson MP,
Pedersen WA, Duan W, Culmsee C, Camandola S. Cellular and molecular mechanisms
underlying perturbed energy metabolism and neuronal degeneration in Alzheimer’s
and Parkinson’s diseases. Ann N Y Acad
Sci 1999;893:154-75.
- 69. Saratikov AS,
Krasnov EA. Chapter III: Stimulative properties of Rhodiola rosea. In: Saratikov AS, Krasnov EA, editors. Rhodiola rosea is a valuable medicinal plant
(Golden Root). Tomsk, Russia:
Tomsk State University; 1987. p. 69-90.
- 70. Panossian A,
Wikman G, Wagner H. Plant adaptogens. III. Earlier and more recent aspects and
concepts on their mode of action. Phytomedicine
1999;6(4):287-300.
- 71. Salnik BU.
Effect of several stimulators on central nervous system energy metabolism
during muscular workload [dissertation]. Tomsk, Russia: Tomsk State Medical
Institute; 1970.
- 72. Adamchuk LB.
Effects of Rhodiola on the process of
energetic recovery of rat under intense muscular workload [dissertation].
Tomsk, Russia: Tomsk State Medical Institute; 1969.
- 73. Danbueva EA.
Effect of stimulators of the central nervous system on lipid metabolism at
different muscular workloads [dissertation]. Tomsk, Russia: Tomsk State Medical
Institute; 1968.
- 74. Revina TA.
Effect of stimulators of the central nervous system on carbohydrate and high
energy phosphorylated compound metabolism in the brain during intense muscular
workload [dissertation] Tomsk, Russia: Tomsk State Medical Institute; 1969.
- 75. Brekhman II,
Dardymov IV. New substances of plant origin which increase non-specific
resistance. Ann Rev Pharmacol
1968;(9):419-30.
- 76. Porsolt RD,
Anton G, Blavet N, Jalfre M. Behavioral despair in rats: a new model sensitive
to antidepressant treatments. Eur J
Pharmacol 1978;47(4):379-91.
- 77. Borsini F,
Meli A. Is the forced swimming test a suitable model for revealing
antidepressant activity? Psychopharmacology
(Berl) 1988;94(2):147-60.
- 78. Willner P.
The validity of animal models of depression. Psychopharmacology (Berl) 1984;83(1):1-16.
- 79. Boon-Niermeijer
EK, van den Berg A, Wikman G, Wiegant FA. Phyto-adaptogens protect against
environmental stress-induced death of embryos from the freshwater snail Lymnaea stagnalis. Phytomedicine 2000;7(5):389-99.
- 80. Lishmanov
IuB, Trifonova ZhV, Tsibin AN, Maslova LV, Dementeva LA. Plasma beta-endorphin
and stress hormones in stress and adaptation. Biull Eksp Biol Med 1987;103(4):422-4.
- 81. Maslova LV,
Kondratev BIu, Maslov LN, Lishmanov IuB. The cardioprotective and
antiadrenergic activity of an extract of Rhodiola
rosea in stress. Eksp Klin Farmakol
1994;57(6):61-3.
- 82. Gerasimova
HD. Effect of Rhodiola rosea extract
on ovarian functional activity. Proc of
Scientific Conference on Endocrinology and Gynecology. Sverdlovsk, Russia.
1970 Sept 15-16. Siberian Branch of the Russian Academy of Sciences. p. 46-8.
83. Saratikov AS, Krasnov EA. Chapter
VI: The influence of Rhodiola on
endocrine glands and the liver. In: Saratikov AS, Krasnov EA, editors. Rhodiola rosea is a valuable medicinal plant
(Golden Root). Tomsk, Russia:
Tomsk State University; 1987. p. 180-93.
- 84. Maimeskulova
LA, Maslov LN, Lishmanov IuB, Krasnov EA. The participation of the mu-, delta-
and kappa-opioid receptors in the realization of the anti-arrhythmia effect of Rhodiola rosea. Eksp Klin Farmakol 1997;60(1):38-9.
- 85. Baranov VB.
The response of cardiovascular system to dosed physical load under the effect
of herbal adaptogen. Contract 93-11-615 Phase I and Phase II. Moscow: Russian
Federation Ministry of Health Institute of Medical and Biological Problems;
1994.
- 86. Bolshakova
IV, Lozovskaia EL, Sapezhinskii II. Antioxidant properties of a series of
extracts from medicinal plants. Biofizika
1997;42(2):480-3.
- 87. Udintsev SN,
Schakhov VP. Decrease of cyclophosphamide haematotoxicity by Rhodiola rosea root extract in mice with
Ehrlich and Lewis transplantable tumors. Eur
J Cancer 1991;27(9):1182.
- 88. Borovskaya
TG, Fomina TI, Iaremenko KV. A decrease in the toxic action of rubomycin on the
small intestine of mice with a transplantable tumor through the use of a
Rhodiola extract. Antiobiot Khimioter
1988;33(8):615-7.
- 89. Udintsev SN,
Krylova SG, Fomina TI. The enhancement of the efficacy of adriamycin by using
hepatoprotectors of plant origin in metastases of Ehrlich’s adenocarcinoma to
the liver in mice. Vopr Onkol
1992;38(10):1217-22.
- 90. Udintsev SN,
Shakhov VP. The role of humoral factors of regenerating liver in the
development of experimental tumors and the effect of Rhodiola rosea extract on this process. Neoplasma 1991;38(3):323-31.
- 91. Salikhova RA,
Aleksandrova IV, Mazurik VK, Mikhailov VF, Ushenkova LN, Poroshenko GG. Effect
of Rhodiola rosea on the yield of
mutation alterations and DNA repair in bone marrow cells. Patol Fiziol Eksp Ter 1997;(4):22-4.
- 92. Bocharova OA,
Matveev BP, Baryshnikov AIu, Figurin KM, Serebriakova RV, Bodrova NB. The
effect of a Rhodiola rosea extract on
the incidence of recurrences of a superficial bladder cancer (experimental
clinical research). Urol Nefrol (Mosk)
1995;(2):46-7.
|