
Issue:
93
Page: 34-41
Adulteration of Skullcap with American Germander
by Steven Foster
HerbalGram.
2012; American Botanical Council
Errata
Photo caption corrections in the printed version of HerbalGram 93 that was made available in February of 2012.
The correct captions are as follows:
Pg. 34: Felty Germander Teucrium polium
Pg. 35: Baikal Skullcap Scutellaria baicalensis
Pg. 39: Felty Germander Teucrium polium
Summary
Skullcap (Scutellaria lateriflora, Lamiaceae), a native American plant, has been used by herbalists and medical practitioners—primarily as a mild nerve sedative—throughout the history of American herbalism. In the early 1980s, it was erroneously implicated as being a possible source of liver toxicity, mostly in combination herbal products. By the early 1990s, it became clear that suspected adulteration or substitution of members of the genus Teucrium were the source of the alleged toxicity that falsely implicated skullcap. Various herbal industry and scientific research groups have published methods for the authentication of skullcap. A paper published in 2011 suggests that the adulteration problem still exists. Various papers relative to the toxicity of Teucrium species and authentication of skullcap are reviewed.
Background
Skullcap, also spelled “scullcap” (Scutellaria lateriflora, Lamiaceae), is a mint family member native to eastern North America that has long been used as a mild nerve sedative or nerve tonic. During the late 19th and 20th centuries, the Eclectic physicians prescribed skullcap for nervousness caused by illness, teething, and mental or physical exhaustion; nervousness with muscular spasms; tremors; and irritability and restlessness with nervous excitability and sleeplessness. Its use as an antispasmodic, nervine tonic, and sedative has continued to the present day. Skullcap fluid extract was listed in the United States Pharmacopeia from 1860 until 1900, and was included in the National Formulary from 1916 to 1942.1
Recent Developments
An article by US Department of Agriculture (USDA) researchers Jianghao Sun and Pei Chen—first published online on July 20, 2011—reported on a laboratory method for identity authentication and quality assessment of dietary supplements labeled as containing skullcap in the journal Annals of Bioanalytical Chemistry.2 The paper described a method of spectral fingerprinting using a liquid chromatography-mass spectrometry (LC-MS) method for authenticating the skullcap products. However, this method is not inexpensive or easily available, which limits its practical utility. The software produces a fingerprint in which different component groups can be discerned by a trained laboratory technician.
From a consumer perspective, the report was disturbing because during the process of developing the analytical method Sun and Chen tested 13 supplement products whose labels stated that the products contained skullcap. However, only five of the products contained S. lateriflora of measurable quality. Three products were found to contain low levels of skullcap-related compounds, and were deemed of low quality. One contained Baikal skullcap (aka Chinese skullcap or huang-qin, S. baicalensis; plant part not specified). Four of the 13 products tested contained the previously known skullcap adulterant Teucrium canadense (Lamiaceae),2 also known as American germander, wild germander, wood sage, wild basil, and in the wholesale herb trade: “pink skullcap.” The presence of T. canadense was primarily based on the detection of verbascoside in the component analysis.
The results of the paper were reported in some news outlets. The problem of adulteration of skullcap commercial supplies with T. canadense or common germander (T. chamaedrys) has been known since the early 1980s, and various efforts have been made to make the problem known to those in the herb trade including publications and notices by the American Botanical Council,1 the American Herbal Pharmacopoeia®,3 and the American Herbal Products Association,4 among others.
Teucrium Toxicity Reports
Various Teucrium species, particularly the common European germander (T. chamaedrys)—widely grown as an evergreen shrub in herb gardens—were associated with several cases of liver toxicity in the early 1990s. The genus Teucrium contains about 250 species. The first modern hint of liver toxicity associated with skullcap adulteration came in an article published in 1981 on alleged hepatotoxicity of mistletoe (Viscum album, Viscaceae), which involved a combination product also containing “skullcap.” At the time, no link was made to hepatotoxicity now associated with skullcap adulteration with Teucrium spp.5 Another red flag was raised in the November 4, 1989 issue of the British Medical Journal. The paper by F. B. MacGregor and colleagues presented 4 case reports of hepatotoxicity associated with the use of herbal products identified as containing “skullcap” and “valerian” (Valeriana officinalis, Caprifoliaceae), in which the authors suggested that “skullcap and valerian are the most likely hepatotoxic components.”6 They also noted that 93 products containing skullcap were available in the United Kingdom at the time,7 and that another paper had reported that a survey of UK wholesalers found that some skullcap available in 1983 was not a Scutellaria species.8 MacGregor and colleagues noted that the situation highlights the problem of accurate formulation of herbal products with properly identified ingredients. They added, “There are no reports to date suggesting any toxic effects from oral ingestion of skullcap.”5
These case reports presented a dilemma on what actually caused the observed liver toxicity. Since oral ingestion of skullcap was not associated with toxicities, could it be that an adulterant to skullcap was the culprit? At the time, the alleged possible toxicity of valerian confounded a number of experts, and seemed remote.
The late pharmacognosist Professor Varro E. Tyler, then executive vice-president of academic affairs at Purdue University and a noted author of pharmacognosy textbooks, in a letter dated August 21, 1992, to ABC Founder and Executive Director Mark Blumenthal, stated: “Personally, I doubt that valerian has any appreciable toxicity when consumed orally. There were a few papers in Germany dating back several years that pointed out that the valepotriates possess an epoxide structure, and they did demonstrate some alkylating activity in cell cultures. Because of this, some thought that the herb must possess potential toxicity. However, those valepotriates decompose rapidly in the stored drug, and even if they were present in the consumed product, they are not readily absorbed. For these reasons, no toxicity has ever been demonstrated in intact animals or human beings, and I believe that there is very little cause for concern. I’ve heard this topic discussed several times at meetings in Germany, and the conclusions always reached were the same as the one stated.”9 Skullcap and valerian were implicated by association despite the suspicion that neither herb elicits the observed hepatotoxicity.
The potential and real toxicity of members of the genus Teucrium gained widespread attention in the early 1990s. Four cases of “skullcap” poisoning, including 1 fatality, were reported from the Riks Hospital in Oslo, Norway’s largest government hospital. The story by reporters Sylvi Leander and Lene Skogstrøm broke in the November 6, 1991 issue of Aftenposten (The Evening Post), Norway’s largest newspaper.10 Once again, at the time it was unclear as to whether S. lateriflora was the offending botanical, or whether possible adulteration with a Teucrium species was the culprit.
A paper published in the July 15, 1992 issue of Annals of Internal Medicine by D. Larrey et al. presented case reports of 7 patients who developed acute hepatitis associated with germander (T. chamaedrys) ingestion. In the 7 patients no other cause of hepatitis was detected. A clear chronological relationship was also established between ingestion of germander and the onset of hepatitis. Liver dysfunction was reversed after use of germander products was discontinued. The mechanism of germander hepatotoxicity could not be determined. Larrey et al. concluded that their study suggests this species of germander may occasionally cause hepatitis.11
Prior to the publication of the above report, the National System of Pharmacovigilance of the French Ministry of Health was aware of the possible problem with germander and had been monitoring it closely. Teucrium chamaedrys, common germander, has long been known as a folk remedy in Europe for the treatment of obesity. Various products were available in Europe, including teas, a medicinal liquor, capsulated products, a product mixed with green tea, as well as bulk herb. The plant had been approved by the French Ministry of Health and Humanitarian Action as an adjuvant in slimming diets, in the symptomatic treatment of mild diarrhea, and as a topical analgesic for oral cavity infections. Products were available both at pharmacies and health-specialty stores. By the spring of 1991, French drug surveillance centers had collected 26 reports of acute hepatitis associated with germander ingestion. All of these cases were benign with an onset delay of 6 weeks to 6 months.12 Finally, in April 1992, the French Ministry of Health prohibited the sale of the herb.13
The Directorate of Pharmacy and Medicines, Ministry of Social Affairs and Integration, Paris, alerted the World Health Organization (WHO) of the toxicity problem on May 15, 1992. Four days later, a communiqué—“Alert No. 27 Herbal Medicines Containing Germander Withdrawn”—was sent by WHO to public health officials.13 In the September 12, 1992 issue of The Lancet, Mostera-Kara et al. yet another fatality from hepatitis after the use of a germander product was reported despite the fact that the manufacturer had withdrawn the product from sale in February of 1991.12 A book series on herb toxicity by DeSmet et al. (1997)14 and the extensive American Herbal Pharmacopoeia and Therapeutic Compendium monograph Skullcap Aerial Parts Scutellaria lateriflora L. by Upton et al. (2009)3 cite several additional reports of T. chamaedrys toxicity cases.
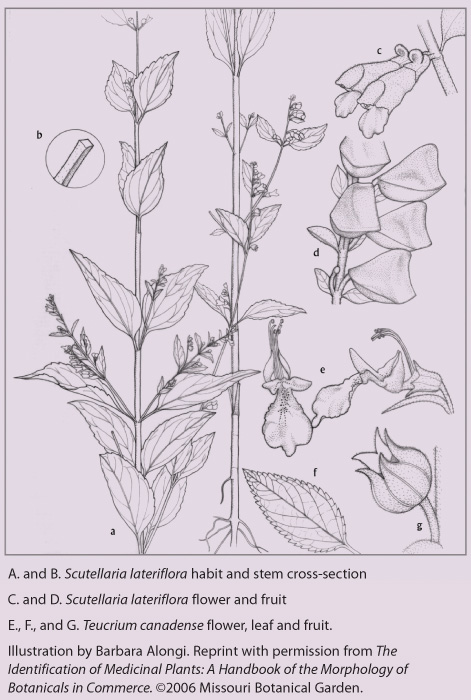 In a October 6, 1992 Memorandum to his superiors, long-time HerbalGram contributor and associate editor Dennis V. C. Awang, PhD, then head of Natural Products, Bureau of Drug Research, Health and Welfare Canada (now Health Canada), noted, “In view of the widely recognized substitution of Teucrium for Scutellaria (Skullcap) I believe that measures must also be taken to ensure the integrity of the skullcap-containing products on the market. The case of hepatotoxicity, reported by MacGregor et al. involving a combination product purported to contain skullcap and valerian, is likely another example of Teucrium (substituted for skullcap) toxicity: no such toxicity has ever been observed with skullcap, and the speculation about possible toxicity due to the alkylating potential of valerian epoxides seems remote, and is without any experimental biological foundation.”15
Despite Dr. Awang’s internal efforts at Health and Welfare Canada to ban Teucrium products from the Canadian market at the time, their availability persisted. A 1996 report in the Canadian Medical Association Journal discussed 2 more case reports of liver disease due to the use of germander products for weight loss.16
The ingestion of germander (T. chamaedrys) had been clearly established as a causative factor in numerous cases of acute hepatitis, and 1 case of fatal hepatic dysfunction. Other Teucrium species have also been implicated. Teucrium polium found in the Mediterranean region was used as a folk remedy for antispasmodic activity and as an antidiabetic. In the course of investigating the herb’s antidiabetic potential, researchers at the University of Jordan observed an unexpected anorexic effect in rats, leading to another rat study that found that a decoction of the herb produced a dose-dependent, reversible anorexic (appetite loss) effect.17 Anorexia from use of T. polium has been linked to liver toxicity and several case reports appear in the literature relative to the hepatotoxicity of T. polium.18,19,20
Skullcap Adulteration – A Persistent Problem
By the early 1990s, it had become clear that any implication of skullcap related to liver toxicity had involved products that were adulterated with Teucrium species. The adulteration and substitution of commercial supplies of S. lateriflora with other species of Scutellaria has been widespread and long-recognized. In the 21st edition of the United States Dispensatory (1926), the authors state, “Scutellaria has been one of the most substituted and adulterated drugs in the Materia Medica. While the S. lateriflora Linné is alone officially recognized as the source of the drug, various other native species of Scutellaria have more frequently represented the article of commerce than the officially specified herb. Chief among these have been S. canescens Nutt. [S. incana subsp. incana] or Western Scullcap, S. cordifolia Muehl. [S. ovata subsp. ovata] or Southern Scullcap, and S. galericulata L.”21 Upton et al. also record S. alpina and S. incana as recently seen skullcap species in the commercial herb supply.3
As has been established, hints that skullcap was adulterated with Teucrium species began to surface in the early 1980s. The 1983 British Herbal Pharmacopoeia monograph contained this note: “Scullcap is frequently adulterated or substituted at source with other species of labiates [i.e., members of the mint family Labiatae, aka Lamiaceae]. Commercial material described is probably derived from a species of Teucrium.”22 According to Roy Upton, executive director of the American Herbal Pharmacopoeia, the description in the 1983 British Herbal Medical Pharmacopoeia actually referred to S. incana.23 The problem was again highlighted in an anonymous brief article in an early-1985 issue of HerbalGram.24 In 1990, this author and James A. Duke, PhD, reported T. canadense to be a widespread adulterant to skullcap.25
Given the fact that an indigenous North American Teucrium species has been implicated as an adulterant to skullcap, as reported in HerbalGram in 1985 and implied in the 1983 British Herbal Pharmacopoeia, the question was raised as to whether or not other species of Teucrium besides T. chamaedrys, especially T. canadensis, may cause acute hepatitis.
For well over a quarter century, T. canadense has entered the wholesale herb trade as “pink skullcap” (or sometimes simply as “skullcap”) a purported adulterant to wholesale supplies of botanical material offered as the herb skullcap. The AHP monograph notes that Teucrium species are characterized by the presence of phenylpropanoid glycosides such as teucrioside in T. chamaedrys and in T. canadense. Verbascoside and teucrioside are the major differentiating glycosides. Also the major flavonoid constituents of S. lateriflora—baicalin, scutellarin, baicalein, wogonin, and chrysin—are not found in the Teucrium adulterants, all of which serve to clearly distinguish the plants using various chemical analytical methods, most of which are provided in the AHP monograph.3 These findings were further supported in a collaborative work between AHP and the United States Department of Agriculture (USDA), which, in a paper by Lin et al. (2009),25 showed that adulteration of S. lateriflora with as little as 1-5% germander herb material could be detected analytically. Additionally, characterization of Scutellaria and T. chamaedrys was provided in analytical papers of Avula et al. 2003,26 Awad et al. 2003,27 Bedir et al. 2003.28
The toxicity of Teucrium spp. is linked to hepatotoxic diterpenes, particularly the neoclerodane diterpene teucrin A. This compound is bioactivated by the cytochrome P450 enzymes in the human liver and gut to create reactive liver-toxic metabolites.2 In contrast, Upton et al. cite several studies providing evidence of a potential hepatoprotective (liver-protective) effect for various skullcap flavones.3
Recent publications such as the report by Sun and Chen (2011) note that the plants are confused because of similar morphology. Sun and Chen write: “Scutellaria lateriflora, commonly known as skullcap, is used as an ingredient in numerous herbal products. However, it has been occasionally adulterated/contaminated with Teucrium canadense and/or Teucrium chamaedrys, commonly known as germander, due to morphological similarities between the two genera.”2 The AHP’s skullcap monograph similarly states: “Skullcap has historically been adulterated with various species of the potential hepatotoxic germander (Teucrium canadense, T. chamaedrys) due to a morphological similarity between S. lateriflora and T. canadense.”3
Many efforts have been made to highlight this adulteration problem and provide clear, reliable information to distinguish between properly identified S. lateriflora and Teucrium species. The most comprehensive and detailed information source on the topic is the 2009 AHP skullcap monograph which includes exhaustive information, illustrations, photographic images, and chromatograms on authentication, morphological differences, and chemical differences, as well as extensive sections on the cultivation, quality control, harvest, and drying of S. lateriflora, with an extensive discussion of adulterants.3 Applequist (2006) also provided clear morphological details to differentiate between S. lateriflora and T. canadense.29 Various papers by S. Gafner et al. also provide detailed information on chemical and microscopic differences between S. lateriflora and T. canadense.30,31 Lin et al. provides further insights and methodology,25 in addition to the recent paper by Sun and Chen.2
Despite the fact that the adulteration problem seems to continue, at least in terms of the products tested by Sun and Chen, many conscientious herbal product manufacturers have made a concerted effort to establish sources of cultivated supply of properly authenticated and labeled S. lateriflora. Various efforts have also been made by horticultural and agricultural researchers to develop production data for S. lateriflora, and methods are reviewed in the comprehensive AHP monograph on skullcap. However, as pointed out in the AHP skullcap monograph, attempts to grow authenticated S. lateriflora have also been thwarted by mislabeling of seeds with S. incana labeled as “S. lateriflora.” This resulted in a commercial grow-out of what the farmer believed to be the correct plant, which was in fact the wrong species.3 This shows the need for due diligence in authentication in all levels of the supply chain.
Conclusion
As pointed out by pharmacologist Ryan Huxtable, PhD, in his editorial in the July 15, 1992 issue of the Annals of Herbal Medicine, the importance of botanical identification of herbal preparations involved in poisoning cases is paramount.32 This adulteration problem has been known for over 25 years, and one would think that the possibility of liability and litigation alone would have prompted this continuing problem to cease, especially in light of the fact that numerous chemical analytical methods have been published, along with simple macroscopic, microscopic, and botanical methods all readily available to laboratories and the herb trade, through Upton’s 2009 AHP monograph and analytical work by the USDA and the University of Mississippi. Persistent, long-standing instances of adulteration and mislabeling of improperly identified botanicals, such as in the instance of skullcap adulteration with T. canadense, must be resolved to ensure that consumers get the herbal products that they expect.
Steven Foster, president of Steven Foster Group Inc., is an author, photographer, and consultant specializing in medicinal and aromatic plants. He is chair of the Board of Trustees of ABC.
This article was peer reviewed by qualified experts in the field of skullcap quality.
Funding for the research, writing, editing, and peer review of this paper was made possible by a series of unrestricted educational grants made to the American Botanical Council for the ABC-AHP-National Center for Natural Products Research Botanical Adulteration Program by a large group of underwriters, most of which are companies that produce, supply, manufacture and/or market herbal ingredients and/or products (current list is posted on the ABC website, www.herbalgram.org and on page 41 of this issue). The author of this paper has no financial relationship with any of these underwriters.
References
- Engels G. Skullcap. HerbalGram. 2009;83:1-2.
- Sun J, Chen P. A flow-injection mass spectrometry fingerprinting method for authentication and quality assessment of Scutellaria lateriflora-based dietary supplements. Analytical and Bioanalytical Chemistry. 2011; 401(5):1281-1588.
- Upton R, ed. Skullcap Aerial Parts Scutellaria lateriflora L. Standards of Analysis, Quality Control and Therapeutics. Scotts Valley, California: American Herbal Pharmacopoeia; 2009.
- American Herbal Products Association. AHPA Guidance Policies; Known Adulterants. Adopted July 1997; revised October 2011. Available at: www.ahpa.org/Default.aspx?tabid=223#section_known_adulterants. Accessed December 14, 2011.
- Harvey J. Colin-Jones DG. Mistletoe Hepatitis. British Medical Journal. 1981;282:186-187.
- MacGregor FB, Abernethy VE, Dahabra S, Cobden I, Hayes PC. Hepatotoxicity of Herbal Remedies. British Medical Journal. 1989;299:1156-1157.
- Phillipson JD, Anderson LA. Herbal remedies used in sedative and anti-rheumatic preparations: part 1. Pharmaceutical Journal 1984; 233:111-144.
- Phillipson JD, Anderson LA. Herbal remedies used in sedative and anti-rheumatic preparations: part 2. Pharmaceutical Journal 1984; 233:80-82.
- Tyler VE. Reference to the Toxicity of Valerian. Letter faxed to Mark Blumenthal. August 21, 1992.
- Leander S, Skogstrom L. Naturmedisin kan gi leverskade. Aftenposten. Oslo, Norway 1991
- Larrey D, Vial T, Pauwels A, et al. Hepatitis after Germander (Teucrium chamaedrys) Administration: Another Instances of Herbal Medicine Hepatotoxicity. Annals of Internal Medicine. 1992;117(2):129-132.
- Mostefa-Kara N, Pauwels A, Pines E, Biour M, Levy VG. Fatal Hepatitis After Herbal Tea. The Lancet. 1992;340:674.
- WHO. Herbal Medicines Containing Germander Withdrawn PHA Information Exchange Service. Alert No. 27. 19 May 1992.
- DeSmet P, Keller K, Hansel R, Chandler R. Adverse Effects of Herbal Drugs. Vol 3. Berlin, Germany: Springer-Verlag; 1997, pp. 137-144.
- Awang D. Toxicity of Teucrium spp. (Germander) and Substitutions for Scutellaria (Skullcap, Scullcap). Memorandum. Dr. K. Bailey D, Bureau of Drug Research, Health and Welfare Canada. Ottawa, Canada October 6, 1992.
- Laliberté L, Villeneuvue JP. Hepatitis after the use of Germander, A Herbal Remedy. Canadian Medical Association. 1996;154(11):1689-1692.
- Gharaibeh MN, H. H. Elayan, and A. S. Salhab. Anorexic Effect of Teucrium polium in Rats. Int. J. Crude Drug Res. 1989;27(4):201-210.
- Mazokopakis E, Lazaridou S, Tzardi M, Mixaki J, Diamantis I, Ganotakis E. Acute cholestatic hepatitis caused by Teucrium polium L. Phytomedicine. 2004;11(1):83-84.
- Mattei A, Rucay P, Samuel D, Feray C, Reynes M, Bismuth H. Liver transplantation for severe acute liver failure after herbal medicine (Teucrium polium) administration. J Hepatol. 1995;22(5):597.
- Savvidou S, Goulis J, Giavazis I, Patsiaoura K, Hytiroglou P, Arvanitakis C. Herb-induced hepatitis by Teucrium polium L.: report of two cases and review of the literature. Eur J Gastroenterol Hepatol. 2007;19(6):507-511.
- Wood HC, LaWall CH, Youngken HW, Anderson JF, Griffith I. The Dispensatory of the United States of America. Philadelphia, PA: J. B. Lippincott Company; 1926. p. 970.
- Scientific Committee of the British Herbal Medicine Association. British Herbal Pharmacopeia. Bournemouth, England: British Herbal Medical Association; 1983. p. 194.
- Upton, R. Personal Communication, December 15, 2011.
- Anon. Scullcap Substitution. HerbalGram. Fall 1985; 2(3):3.
- Lin LZ, Harnly JM, Upton R. Comparison of the phenolic component profiles of skullcap (Scutellaria lateriflora) and germander (Teucrium canadense and T. chamaedrys), a potentially hepatotoxic adulterant. Phytochem Anal. 2009;20(4):298-306.
- Avula B, Manyam RB, Bedir E, Khan IA. Rapid separation and determination of four phenylpropanoid glycosides from T. chamaedrys by capillary electrophoresis method. Chromatographia 2003; 58:751-755.
- Awad R, Arnason JT, Trudeau V, Bergeron C, Budzinski JW, Foster BC, Merali Z. Phytochemical and biological analysis of skullcap (Scutellaria lateriflora L.): a medicinal plant with anxiolytic properties. Phytomedicine. 2003;10:640-649.
- Bedir E, Manyam R, Khan IA. Neo-clerodane diterpenoids and phenlethanoid glycosides from Teucrium chamaedrys L. Phytochemistry 2003; 63:977-983.
- Applequist W. The Identification of Medicinal Plants: A Handbook of the Morphology of Botanicals in Commerce. St. Louis, MO, and Austin, TX: Missouri Botanical Garden and the American Botanical Council 2006. pp. 147-149.
- Gafner S, Sudberg S, Sudberg ÉM, Bergeron C, Batcha LL, Guinaudeau H, Gauthier R, Angerhofer CK. Analysis of Scutellaria lateriflora and its adulterants Teucrium canadense and Teucrium chamaedrys by LC-UV/MS, TLC, and digital photomicroscopy. J AOAC Int. 2003;86(3):453-460.
- Gafner S, Reich J, Bergeron C, Smith J, Batcha LL, Angerhofer CK. Comparison of Different Extracts of Scutellaria lateriflora L. by HPLC.
|