
Issue:
94
Page: 62-66
The Adulteration of Commercial “Grapefruit Seed Extract” with Synthetic Antimicrobial and Disinfectant Compounds
by John H. Cardellina II, PhD
HerbalGram.
2012; American Botanical Council
Material called “grapefruit seed extract” (GFSE1) has been sold in the natural products market for 3 decades or more as an ingredient in or preservative for cosmetic and dermatological preparations and also in dietary supplements. GFSE, supposedly an extract of the seeds of the common grapefruit (Citrus x paradisi, Rutaceae), has been touted in popular literature as a natural antimicrobial agent for both topical and internal use, including, but not limited to, eczema, acne, cold sores, athlete’s foot, sore throats, thrush, vaginal infections, colds, various gastrointestinal disorders and infections, allergies, and gingivitis.2,3 Much of the commercially available GFSE is produced via proprietary methods that purportedly involve the use of catalytic processes and the addition of solvents and/or other chemicals. For example, in the case of one of the leading branded consumer products labeled as containing “grapefruit seed extract,” this process has not been fully disclosed or explained in any publicly available literature, but is claimed to involve a multistep process that includes boiling ground, dried seeds and pulp in water, then “…distillation, catalytic conversion and ammoniation…”4 to yield GFSE, the active ingredient of which “…is a quaternary ammonium chloride (a diphenol hydroxybenzene reacted with ammonium chloride) similar to benzethonium chloride…”.4
In 1991, a collaboration led by Nishina (Food Research Laboratory, Nippon Oil and Fats Co.; Tokyo, Japan) published the first analysis of commercial GFSE and reported that preparative high-performance liquid chromatography (HPLC) led to identification of methyl p-hydroxybenzoate, a preservative, and triclosan, a microbicide and disinfectant.5 Five years later, Sakamoto et al., at the Japanese National Institute of Health Sciences in Tokyo, repeated the analysis of GFSE using HPLC-ESIMS (HPLC with electrospray ionization mass spectrometry), and compared the commercial GFSE with ethanolic extracts of grapefruit seeds that they prepared themselves.6 Like the Nishina et al. study, Sakamoto found methyl p-hydroxybenzoate and triclosan as 1.66% and 1.97% of the GFSE, respectively; perhaps more importantly, no trace of these compounds was found in their ethanolic extract of grapefruit seeds. Unfortunately, these 2 important papers may not have received the level of attention they deserved because they were published in Japanese-language journals and went largely unnoticed outside of Japan.
However, at least 2 groups did take note of Sakamoto’s report. The von Woedtke group (Ernst Moritz Amdt University; Greifswald, Germany) analyzed 6 commercially available GFSE extracts; five of them exhibited significant antimicrobial activity. The 5 antimicrobial GFSE extracts were found by TLC (thin-layer chromatography) to contain the synthetic microbicide benzethonium chloride; three of those also contained triclosan and methyl p-hydroxybenzoate.7 The one remaining GFSE product and fresh extracts prepared from grapefruit seeds with glycerol, water, ethanol, and combinations of the three, contained none of the 3 synthetic preservatives/disinfectants, nor did any of these samples exhibit any antimicrobial activity. Thus, there was a direct correlation of the presence of synthetic preservatives, disinfectants, and microbicides with the antimicrobial activity observed in the GFSE products.
Takeoka’s group at the USDA (Western Regional Research Center; Albany, California) subsequently published 2 analyses of GFSE formulations.8,9 In the first, they analyzed what the authors noted to be two of the leading commercial GFSE products in the United States, one a liquid concentrate and the other a concentrated powder, by a combination of extraction, HPLC-ultraviolet spectroscopy (HPLC-UV), mass spectrometry (MS), and nuclear magnetic resonance spectroscopy (NMR). Rather than methyl p-hydroxybenzoate and triclosan, they found benzethonium chloride as 8% of the mass of the liquid GFSE sample. Benzethonium chloride was also found in the concentrated powder, but was not quantified. Later, Takeoka et al.9 examined the contents of the same commercial product analyzed by Nishina et al.5 by HPLC combined with ESIMS and ESIMS/MS, but they found no triclosan, methyl p-hydroxybenzoate, or benzethonium chloride. Instead, they found a mixture of benzalkonium chlorides as an astonishing 22% of the extract weight. These studies prompted even broader surveys of GFSE preparations and products containing them.
Avula et al. developed an HPLC-UV-MS method for the simultaneous identification and quantification of benzethonium chloride, methyl p-hydroxybenzoate, and triclosan in GFSE products and used it to evaluate 9 unidentified commercial GFSE products, one pomegranate (Punica granatum, Punicaceae) seed extract, and a freshly prepared methanolic extract of grapefruit seeds.10 No methyl p-hydroxybenzoate was detected in any sample; triclosan was detected in 3 of the commercial GFSE preparations (0.009-1.13%); and benzethonium chloride was found in 8 of 9 commercial GFSE preparations—one below the limit of quantification, one at 0.29%, and the other six at 5.97-21.84% of the mass. The commercial pomegranate seed extract (used as a control) and the fresh grapefruit seed extract were devoid of all 3 compounds.
Ganzera et al. (University of Innsbruck, Austria) developed and validated an HPLC-UV-MS method for simultaneous determination of 18 possible preservatives, disinfectants, and microbicides in GFSE and tested 9 unidentified commercial products used for eco-farming—8 liquids and 1 powder.11 All the products except 1 liquid claimed GFSE as a component of the product. The non-GFSE product contained none of the 18 possible contaminants, but 6 of the remaining 7 products contained between one and five of the analytes (Table 1). The liquid GFSE preparation contained methyl p-hydroxybenzoate (0.31%), benzoic acid (0.13%), methyl benzoate (0.05%), propyl p-hydroxybenzoate (0.12%), C12 benzalkonium chloride (3.47%), C14 benzalkonium chloride (1.15%), and C16 benzalkonium chloride (0.31%).
 Spinosi et al. (Istituto Zooprofilattico Sperimentale dell’ Abruzzo e del Molise ‘G. Caporale’; Teramo, Italia) used gas chromatography-mass spectrometry (GC-MS) to examine 17 GFSE products used as “organic” treatments of diseases of honey bees and found benzethonium chloride (0.003-21.5% in 14 products), cetrimonium bromide (3.202-11.656% in 5 products), and decytrimethylammonium chloride (10.32% in 1 product).12 Their results suggested a strong correlation between the presence of chemical additives (disinfectants) and observed antimicrobial effects.
Sugimoto et al. (National Institute of Health Sciences; Tokyo, Japan) used HPLC-MS and 1H-NMR to analyze 13 GFSE products used as food additives from 6 manufacturers, 5 dietary supplements from 4 manufacturers, 16 cosmetic products from 10 manufacturers, and 7 disinfectant or deodorant sprays from 7 manufacturers for the presence of the synthetic disinfectants benzethonium and benzalkonium chlorides.13 Twelve of the food additive GFSE products contained one of the synthetic disinfectants—11 contained benzalkonium chloride and one contained benzethonium chloride (39.06%). The 3 concentrated liquid products with benzalkonium chloride contained 12.156-13.898% of the disinfectant; the other 8 products were dilutions or formulations made with one of the concentrated liquids containing 0.007-1.739% benzalkonium chloride. Two of the dietary supplements contained neither of the synthetic disinfectants, while the other three had benzethonium chloride in 0.008%, 0.215%, and 6.78 %, respectively. All of the cosmetic products contained benzethonium chloride (14.596-28.826%); one of them also contained 3.456% benzalkonium chloride. All of the disinfectant/deodorant sprays contained either benzalkonium chloride (0.014-0.208%) or benzethonium chloride (0.019-0.260%).
Bekiroglu et al. (Medical Products Agency; Uppsala, Sweden) subsequently developed and validated an NMR method for the quantitative analysis of benzethonium chloride in GFSE; they analyzed 3 unidentified commercial samples of GFSE and found 78 mg/mL of benzethonium chloride.14
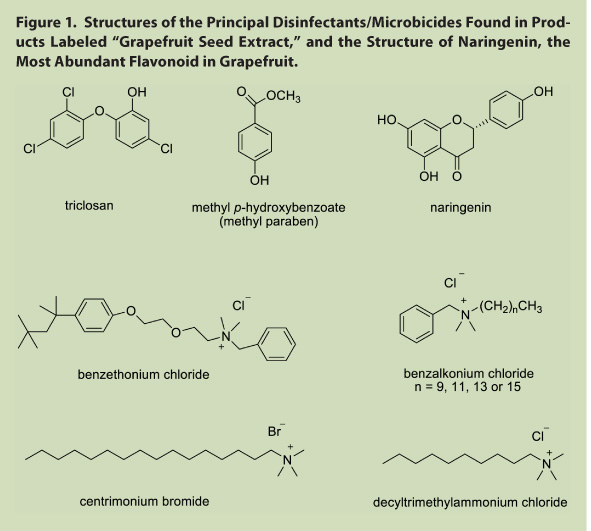
The structures of the synthetic disinfectants and microbicides discussed herein are shown in Figure 1, along with that of naringenin, a flavonoid abundant in grapefruit.
 In addition to the scientific studies cited above, the American Herbal Products Association (AHPA), a trade association representing the herbal industry in the United States, has repeatedly worked to draw attention to the problem of adulterated GFSE via communications regarding recent GFSE publications to its industry members.15,16,17 Further, Caldecott has published an essay on the subject, with emphasis on potential safety concerns about the adulterants that have been reported in commercial GFSE products.18 To this author’s knowledge, and based on extensive literature searches, there is no record of the medicinal use of grapefruit seed or grapefruit seed extract in any ancient or modern treatise on traditional herbal medicine, modern pharmacopeia, or other compilation of medicinal plants from any culture. A search of the PubMed database at the United States National Library of Medicine under the search term “grapefruit seed extract AND clinical trials” yielded no search results. [Search of www.pubmed.org, March 21, 2012.] The PubMed database, and others, do contain citations and abstracts of published reports on GFSE antimicrobial activity in experimental (in vitro) and in vivo animal models, but because the actual composition of such GFSE materials used in these studies was not clarified by accompanying chemical analyses, this entire body of research is questionable.
Conclusion
A significant amount, and possibly a majority, of ingredients, dietary supplements and/or cosmetics labeled as or containing grapefruit seed extract (GFSE) is adulterated, and any observed antimicrobial activity is due to synthetic additives, not the grapefruit seed extract itself. Tests conducted in multiple laboratories over almost 20 years indicated that all commercial GFSE preparations that exhibited antimicrobial activity contained one or more synthetic microbicides/disinfectants, while freshly-prepared extracts of grapefruit seeds made with a variety of extraction solvents neither exhibited antimicrobial activity nor contained the antimicrobial synthetic compounds found in the commercial ingredient materials. Furthermore, over the course of the 18 years covered by the various analyses, the actual antimicrobial compounds found in the putative grapefruit seed extracts changed from triclosan and methyl p-hydroxybenzoate in early samples to benzethonium chloride in the middle years to mixtures of benzalkonium and/or alkonium chlorides in more recent years. The suggestion on a commercial website4 that these antimicrobial compounds are formed from the phenolic compounds naturally occurring in grapefruit seed and pulp by heating them with water, ammonium chloride, and hydrochloric acid is not supported by chemical evidence, or any known organic chemistry pathway. None of these compounds could be formed from flavonoids like naringenin, the most abundant flavonoid in grapefruit seeds, pulp, and peel, or other grapefruit seed constituents (e.g., limonoids) and ammonium chloride; the alkyl chains and substituent arrays found in the antimicrobial adulterants are not naturally present in grapefruit seed and cannot be prepared from those materials. The fact that the antimicrobial components found in GFSE changed from 1991 to 2008 not only argues against such in situ synthesis (i.e., occurring naturally or synthesized in the processing of grapefruit seed material itself), but is suggestive of efforts by manufacturers of these commercial materials to stay one step ahead of analytical methods to detect adulteration.
Safety Notes: A Swedish team has reported that a female (age not provided) on long-term warfarin (coumadin) treatment with regular monitoring consumed several drops of GFSE for 3 days. Three days later she developed a minor subcutaneous hematoma, at which time her INR (international normalized ratio, a measure of blood coagulation time) was found to be 7.9 (normal range for warfarin patients is 1-3). NMR analysis of the GFSE product she had been taking revealed the presence of water, glycerol, and benzethonium chloride. Benzethonium chloride was tested and found to be a potent inhibitor of the metabolizing enzymes CYP3A4 and CYP2C9. Inhibition of these enzymes results in prolonged, higher levels of circulating coumadin, thus increasing clotting times. The investigators (Medical Products Agency; Uppsala, Sweden) thus concluded that benzethonium chloride in the GFSE product consumed by the patient was responsible for the spike in her INR.19
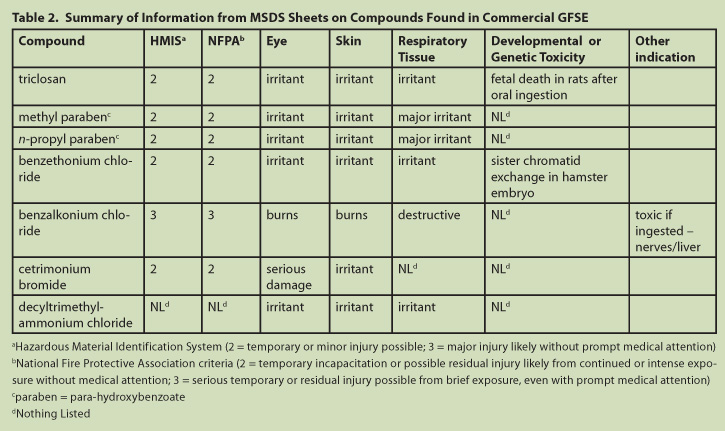
A review of the Material Safety Data Sheets (MSDS) for the synthetic disinfectants and microbicides found in commercial samples of GFSE revealed a general pattern of eye, skin, and respiratory tract irritation, a few indications of genetic, developmental, or organ toxicity, and a moderate health hazard, although a few citations of more serious adverse effects were noted. A summary is provided in Table 2, below.
 John H. Cardellina II, PhD, is a natural product, organic, medicinal, and analytical chemist who has held positions in the pharmaceutical industry, academia, the National Cancer Institute, Office of Dietary Supplements, the US Army Research Institute of Infectious Diseases, and the Council for Responsible Nutrition. He is also a consultant in many areas of natural products research. Dr. Cardellina is the author or co-author of nearly 200 publications and co-inventor on numerous patents covering 13 classes of natural products and synthetic compounds. Among many organizational positions he has held, he is a past-president of the American Society of Pharmacognosy, a longtime member of the Advisory Board of the American Botanical Council, current Chair of the Board of Directors of the American Herbal Pharmacopoeia, Chair of the American Society of Pharmacognosy Foundation, and the book review editor for the Journal of Natural Products.
Acknowledgements
The author is deeply grateful to S. Matsunaga, PhD, for translating references 5, 6, and 13, and to S. Cardellina, MBA, for assistance with the translation of reference 12.
This article was peer reviewed by qualified experts in analytical chemistry and related disciplines, including some of the researchers whose papers are summarized and cited in this review.
Funding for the research, writing, editing, and peer review of this paper was made possible by a series of unrestricted educational grants made to the American Botanical Council for the ABC-AHP-NCNPR Botanical Adulteration Program. ABC, the American Herbal Pharmacopoeia, and the University of Mississippi’s National Center on Natural Products Research are grateful for the support from a large group of underwriters, most of which are trade associations and companies that produce, supply, manufacture, and/or market herbal ingredients and/or products (current list is posted on the ABC website, www.herbalgram.org). The author of this review paper has no financial relationship with any of these underwriters.
References and Notes
- Some of the papers cited in this article use the acronym GSE to refer to grapefruit seed extract. However, the author of this article prefers using the acronym GFSE for grapefruit seed extract to differentiate it from, and avoid confusion with, grape seed extract (from Vitis vinifera, Vitaceae), a widely used botanical product that is usually referred to as GSE. In an example of this potential (now realized) confusion, the authors of a recent paper on the pharmacological activity of grapefruit seed extract (Cao S, et al. A mitochondria-dependent pathway mediates the apoptosis of GSE-induced yeast. PLoS ONE 2012. 7: e32943) describe grapefruit seed extract as containing catechins and procyanidins, citing a reference to grape seed chemistry.
- Sharamon S, Baginski BJ. The Healing Power of Grapefruit Seed. Twin Lakes, Wisconsin: Lotus Light Publications; 1997.
- Sachs A. The Authoritative Guide to Grapefruit Seed Extracts. Mendocino, California: LifeRhythm; 1997.
- Nutriteam.com. Citricidal Grapefruit seed extract. Available at: www.nutriteam.com/citricidal.htm. Accessed September 28, 2011.
- Nishina A, Kihara H, Uchibori T, Oi T. Antimicrobial substances in“DF-100”, extract of grapefruit seeds. Bokin Bobai (J. Antibact Antifung Agents) 1991;19:401-404. [commercial product identified in the paper]
- Sakamoto S, Sato K, Maitani T, Yamada, T. Analysis of components in natural food additive “grapefruit seed extract” by HPLC and LC/MS. Eisei Shikenjo Hokoku (Bull Natl Inst Health Sci.). 1996;114:38-42.
- von Woedtke T, Schlüter B, Pflegel P, Lindequist U, Jülich W.-D. Aspects of the antimicrobial efficacy of grapefruit seed extract and its relation to preservative substances contained. Pharmazie;1999. 54:452-456. [commercial products identified]
- Takeoka G, Lan D, Wong RY, Lundin R, Mahoney N. Identification of benzethonium chloride in commercial grapefruit seed extracts. J Agric Food Chem. 2001;49:3316-3320. [commercial products identified]
- Takeoka GR, Dao LT, Wong RY, Harden LA. Identification of benzalkonium chloride in commercial grapefruit seed extracts. J Agric Food Chem. 2005;53:7630-7636. [commercial products identified]
- Avula B, Dentali S, Khan IA. Simultaneous identification and quantification by liquid chromatography of benzethonium chloride, methyl paraben and triclosan in commercial products labeled as grapefruit seed extract. Pharmazie. 2007;62:593-596. Reference samples of all specimens tested are deposited at the Institute of Pharmacy, University of Innsbruck, Innsbruck, Austria.
- Ganzera M, Aberham A, Stuppner H. Development and validation of an HPLC/UV/MS method for simultaneous determination of 18 preservatives in grapefruit seed extract. J Agric Food Chem. 2006;54:3768-3772.
- Spinosi V, Semprini P, Langella V. Presence of chemical additives and microbial inhibition capacity in grapefruit seed extracts used in apiculture. Veterinaria Italiana. 2007;43:109-113.
- Sugimoto N, Tada A, Kuroyanagi M, Yoneda Y, Yun YS, Kunugi A, Sato K, Yamazaki T, Tanamoto K. Survey of synthetic disinfectants in grapefruit seed extract and its compounded products. Shokuhin Eiseigaku Zasshi. 2008;49:56-62.
- Bekiroglu S, Myrberg O, Ostman K, Ek M, Arvidsson T, Rundlöf, T, Hakkarainen B. Validation of a quantitative NMR method for suspected counterfeit products exemplified on determination of benzethonium chloride in grapefruit seed extracts. J Pharm Biomed Anal 2008;47:958-961.
- Anon. Antiinfective compounds in grapefruit seed extract – not natural. AHPA Report. 2005; 20(7):12.
- Anon. Adulteration of commercial GSE with synthetic preservative: nailed again. AHPA Report. 2005;20(9):10.
- Anon. Grapefruit seed extract times three – the title says it all. AHPA Report. 2006;21(6):11.
- Caldecott T. Grapefruit seed extract. Medical Herbalism. 2005;14:1-2.
- Brandin J, Myrberg O, Rundlöf T, Arvidsson A-K, Brenning G. Adverse effects by grapefruit seed extract products in patients on warfarin therapy. Eur J Clin Pharmacol. 2007;63:565-570.
|